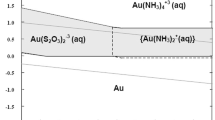
Abstract
The dissolution of gold in alkaline solutions containing cyanide ions is of great importance and its application in the MacArthur-Forrest process towards the end of the last century revolutionized the extraction of gold from its ores. Because of this, and its use in many industrial processes involving gold, it has been widely studied. What is known — and still unknown — about this reaction constitutes the main theme of this second instalment of a review of the anodic behaviour of gold.
Similar content being viewed by others
References
M. J. Nicol, Gold Bull., 1980, 13,(2), 46–55
R. Thacker and T. P. Hoare, Electrochem. Technol., 1964, 2, 61–64
S. E. S. el Wakkad and A. M. Shams el Din, J. Chem. Soc., 1954, 3098–3102
K. J. Cathro and D. F. A. Koch, J. Electrochem. Soc., 1964, 111, 1416–1420
R. Cordova, M. E. Martins and A. J. Arvia, J. Electrochem. Soc., 1979, 126, 1172–1176
V. Kudryk and H. Kellogg, J. Met., 1954, 6, 541–548
D. W. Kirk, F. R. Foulkes and W. F. Graydon, J. Electrochem Soc., 1978, 125, 1436–1443
D. W. Kirk, F. R. Foulkes, and W. F. Graydon, J. Electrochem. Soc., 1979, 126, 2287–2288
E. T. Eisenmann, J. Electrochem. Soc., 1978, 125, 717–723
D. M. MacArthur, J. Electrochem. Soc., 1972, 119, 672–676
T. P. Pan and C. C. Wan, J. Appl. Electrochem., 1979, 9, 653–655
K. J. Cathro, Proc. Aust. lnst. Min. Metall., 1963, 207, 181–205
T. Mills, Ph.D. Thesis, University of Melbourne, 1951
M. J. Nicol, E. Schalch, P. Balestra and H. Hegedus, J.S. Afr. Inst. Min. Metall., 1979, 79, 191–198
J. D. E. Mcintyre and W. F. Peck, J. Electrochem. Soc., 1976, 123, 1800–1813
C. B. Munier, Plating, 1969, 56, 1151–1157
N.J. Louw, A. M. Edwards and H. W. Gussman, Chemsa, 1977, 3, 135–136
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nicol, M.J. The anodic behaviour of gold. Gold Bull 13, 105–111 (1980). https://doi.org/10.1007/BF03216548
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03216548