Summary
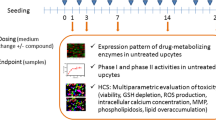
Over past decades, numerous in vitro and/or ex vivo models have been developped to investigate drug metabolism. In the order of complexity we found the isolated perfused liver, hepatocytes in co-culture with epithelial cells, hepatocytes in suspension and in primary culture and subcellular hepatic microsomal fractions. Because they can be easily prepared from both animals (pharmacological and toxicological species) and humans (whole livers as well as biopsies obtained during surgery) hepatocytes in primary culture provide the most powerfull model to better elucidate drug behavior at an early stage of preclinical develompent such as: the characterization of main biotransformation reactions, the identification of phase I and phase II isozymes involved in such reactions, the evaluation of inter-species differences allowing the selection of a second toxicological animal species more closely related to man on the basis of metabolic profiles, the detection of the inducing and/or inhibitory effects of a drug on metabolic enzymes, the prediction of drug interactions, the estimation of inter-individual variability in biotransformation reactions. The use of hepatocytes, and in particular those obtained from humans, at an early stage of drug development allows the obtention of more predictive preclinical data and a better knowledge of drug behavior in humans before the first administration of the drug in healthy volunteers.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J.F. Le Bigot, J.M. Begue, J.R. Kiechel and A. Guillouzo (1987). Species differences in metabolism of ketotifen in rat, rabbit and man: demonstration of similar pathways in vivo and in cultured hepatocytes. Life Sci. 40: 883–891
R. Rahmani, B. Richard, G. Fabre and J.P. Cano (1988). Extrapolation of preclinical pharmacokinetic data to therapeutic drug use. Xenobiotica 18: 71–86
B. Richard, G. Fabre, G. de Souza and J.P. Cano (1987). Metabolism of mitoxantrone by hepatocytes in primary culture isolated from different species including man. Proc. Am. Ass. Cancer Res. 28: 1674
C.E. Green, S.E. Le Valley and C.A. Tyson (1986). Comparison of amphetamine metabolism using isolated hepatocytes from five species including human J. Pharm. Exp. Ther. 237: 931–937
R.J. Chenery, A. Ayrton, H.G. Oldham, P. Standring, S.J. Norman, T. Seddon and R. Kirby (1987). Diazepam metabolism in cultured hepatocytes from rat, rabbit, dog, guinea pig and man Drug Metab. Dispos. 15: 312–318
M.H. Grant, M.D. Burke, G.M. Hawksworth, S.J. Duthie, J. Engeset and J.C. Petrie (1987). Human adult hepatocytes in primary monolayer culture. Maintenance of mixed function oxidase and conjugation pathways of drug metabolism. Biochem. Pharmacol. 36:2311–2316
J.P. Cano, G. Fabre, P. Maurel, N. Bichet, Y. Berger and P. Vic (1988). Inter-individual variability and induction of cytochromes P450 and UDP-glucuronosyl transferases in human liver microsomes and primary cultures of human hepatocytes in: Cellular and Molecular aspects of Glucuronidation (eds. G. Siest, J. Magdalou, B. Burchell) INSERM/John Libbey Eurotext Ltd, Vol 173, pp 249–260
P. Heizmann, M. Eckert and W.H. Ziegler (1983). Pharmacokinetics and bioavailability of midazolam in man Br. J. Clin. Pharmacol. 16:43S-49S
P. Crevat-Pisano, S. Dragna, C. Granthil, P. Coassolo, J.P. Cano and G. Francois (1986). Plasma concentrations and pharmacokinetics of midazolam during anesthesia. J. Pharm. Pharmac., 38:578
G. Fabre, R. Rahmani, M. Placidi, J. Combalbert, J. Covo, J.P. Cano, C. Coulange, M. Ducros and M. Rampai (1988). Characterization of midazolam metabolism using human hepatic microsomal fractions and hepatocytes in suspension obtained by perfusing whole human livers. Biochem. Pharmacol. 37:4389
G. Fabre, P. Crevat-Pisano, S. Dragna, J. Covo, Y. Barra and J.P. Cano (1988). Involvement of the macrolidc antibiotic inducible cytochrome P450 LM3C in the metabolism of midazolam by microsomal fractions prepared from rabbit liver. Biochem. Pharmacol. 37:1947–1953
H.R. Ochs, D.J. Greenblatt, W. Eichelkraut, C. Bakker, R. Göbel and N. Hahn (1987). Hepatic vs. gastrointestinal presystemic extraction of oral midazolam and flurazepam J. Pharm. Exp. Ther. 243: 852–856
P. Bertault-Peres, C. Bonfils, G. Fabre, S. Just, J.P. Cano and P. Maurel (1987). Metabolism of Cyclosporin AH Implication of macrolide antibiotic inducible cytochrome P4503C from rabbit liver microsomes Drug Metab. Dispos. 15: 391–398
J. Combalbert, I. Fabre, G. Fabre, I. Dalet, J. Derancourt, J.P. Cano and P. Maurel (1989). Metabolism of Cyclosporin AIV Purification and identification of the rifampicin inducible human liver cytochrome P450 (Cyclosporin A oxidase) as a product of P450IIIA gene subfamily. Drug Metab. Dispos. 17: 197–207
N.K. Wadhwa, T.J. Schroeder, A.J. Pesce, S.A. Myre, C.W. Chardy and M.R. First (1987). Ciclosporine drug interactions. A review Ther. Drug. Monit. 9:399
K.W. Bock, J. Witfang, R. Blume, D. Ullrich and J. Bircher (1987). Paracetamol as a test drug to determine glucuronide formation in man. Effects of inducers and of smoking. Eur. J. Clin. Pharmacol. 31:677–683
K.W. Bock and B.S. Bock-Henning (1987). Differential induction of human liver UDP-glucurono- syltransferase activities by phenobarbital — type inducers Biochem. Pharmacol. 36: 4137–4143
A.R. Boobis, M.J. Brodie, G.C Kahn, D.R. Fletcher, J.H. Saunders and D.S. Davis (1980). Monooxygenase activity of human liver in microsomal fractions of needle biopsy specimens. Br. J. Clin. Pharmac. 9:11–19
O. Pelkonen, M. Pasanen, M. Kuha, B.J. Gachalyi, M. Kairaluoma, E.A. Sotianemi, S.S. Park, F.K. Friedman and H.V. Gelboin (1986). The effect of cigarette smoking on 7-ethoxyresorufin O- deethylase and other monooxygenase activities in human liver: analyses with monoclonal antibodies. Br. J. Clin. Pharmacol. 22:125–129
P. Maurel, I. Dalet-Beluche, C. Dalet, C. Bonfils, P. Bories, P. Bautet and H. Michel (1986). Activités enzymatiques et dosage immunologique d’une forme de cytochrome P-450 microsomal dans des biopsies hépatiques à l’aiguille in: Hepatotoxicity of drugs (ed. J.P. Fillastre) pp. 289–304
D.N. Nebert, D.R. Nelson, M. Adesnik, M.J. Coon, R.N. Estabrook, F.J. Gonzalez, F.P. Guengerich, I.C. Gunsalus, E.F. Johnson, B. Kemper, W. Levin, I.R. Phillips, R. Sato and M.R. Waterman (1989). The P450 Superfamiry: Update listing of all genes and recommended nomenclature for the chromosomal loci D.N.A. vol 8, No 1:1–13
P.B. Watkins, S.A. Wrighton, P. Maurel, E.G. Schuetz, G. Mendez-Picon, G.A. Parker and P.S. Guzelian (1985). Identification of an inducible form of cytochrome P-450 in human liver Proc. Natl. Acad. Sei. U.S.A. 82: 6310–6314
M.S. Lennard, L.E. Ramsay, J.H. Silas, G.T. Tucker and H.F. Woods (1983). Protecting the poor metabolizer. Clinical consequences of genetic polymorphism of drug oxidation Pharm. Int., 4:53–57
A. Kupfer and P. Preisig (1984). Pharmacogenetics of mephenytoin. A new drug hydroxylation polymorphism in man. Eur. J. Clin. Pharmacol. 26: 753–759
F.P. Guengerich, P.H. Beaune, D.R. Umbenhauer, P.F. Chur chill, R.W. Bork, G.A. Dannan, R.G. Knodell, R.S. Lloyd and M.V. Martin (1987). Cytochrome P-450 enzymes involved in genetic polymorphism of drug oxidation in man. Biochem. Soc. Trans. 15:576–578
J.P. Cano, Y. Berger and G. Fabrc (1990). Relevance of genetic polymorphism in metabolism for drug development: industrial point of view, in: “Consensus Conference on Pharmacogenetics” ed. P. Bechtel. In press
G. Fabre, I. Fabre, D.A. Gewirtz and J.D. Goldman (1985). Characteristics of the formation and membrane transport of 7-hydroxymethotexate in freshly isolated rabbit hepatocytes. Cancer Res. 45:1086–1091
I. Fabre, G. Fabre, P. Maurel, P. Berthault-Peres and J.P. Cano (1988). Metabolism of cyclosporin A: III. Interaction of the macrolide antibiotic, erythromycin, using rabbit hepatocytes and microsomal fractions. Drug Metab. Dispos. 16:296–301
L, Pichard, I. Fabre, G. Fabre, J. Domergue, B. Saint-Aubert, G. Mourad and P. Maurel (1990). Cyclosporin A. drug interactions. Screening for inducers and inhibitors of Cytochrome P450 (Cyclosporin A oxidase) in primary cultures of human hepatocytes and in liver microsomes. Drug Metab. Dispos. 18, in press
Ch. Briot, G. Fabre, E. Marti, M. Bourrie, B. Saint-Aubert, H. Joyeux, Y. Berger and J.P. Cano (1990). Glucuronidation of valproic acid by both human liver microsomal fractions and human hepatocytes in primary cultures. Xenobiotica, manuscript in preparation
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fabre, G., Combalbert, J., Berger, Y. et al. Human hepatocytes as a keyin vitro model to improve preclinical drug development. Eur. J. Drug Metab. Pharmacokinet. 15, 165–171 (1990). https://doi.org/10.1007/BF03190200
Issue Date:
DOI: https://doi.org/10.1007/BF03190200