Summary
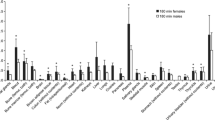
The study carried out on rats given orally then-hexane lipido/sterolic extract ofSerenoa repens (LSESR), supplemented with [14C]-labelled oleic or lauric acids or β-sitosterol, demonstrated that radioactivity uptake in prostatic tissues shows the highest level in th case of administration of LSESR supplemented with [14C]-labelled oleic acid. This was clearly demonstrated on a rat with an induced fibro-muscular hyperplasia of the prostate and by quantitative measurements of radioactivity. Ratios of radioactivity in tissues compared to plasma show an uptake of radioactivity greater in prostate as compared to other genital organs, i.e. the seminal vesicles or to other organs such as liver.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Neuzil E., Cousse H. (1993): Le palmier-scieSerenoa repens: aspects botaniques et chimiques. Bull. Soc. Pharm. Bordeaux, 132, 121–141.
Small J.K. (1926): The saw-palmettoSerenoa repens, J. NY Bot. Gard., 27, 193–202.
Sherrard C.C. (1894): Saw palmetto (Sabal serrulata). Proc. Am. Pharm. Assoc., 42, 309–315.
Kraemer H. (1907): A text-book of botany and pharmacognosy, vol. 1, 2nd edn. Philadelphia: J.B. Lippincott.
Maisch V.M. (1899): A manual of organic materia, vol. 1, 7th edn. Philadelphia: Lea Brother.
Sayre L.E. (1907): A manual of organic materia and pharmacology, vol. A, 3rd edn. Philadelphia: P. Blakinston’s & Sons.
Shoemaker J.V. (1896): A practical treatise on materia medica and therapeutics with special reference to the clinical application of drugs. 2 vols, 4th edn. Philadelphia: Davis.
British Pharmaceutical Codex, vol. 1. (1923): 959.
National Formulary. vol. 1, Am. Pharm. Assoc., 7th edn, pp 137; 8th edn, pp 457–459.
Authie D., Cauquil J. (1987): Appréciation de l’efficacité de Permixon en pratique quotidienne. CR Thérap. Pharmacol. Clin., 5.
Champault G., Patel J.C., Bonnard A.M. (1984): A double-blind trial of an extract of the plantSerenoa repens in benign prostatic hyperplasia, Br. J. Clin. Pharmacol., 18, 461–462.
Cukier J., Ducassou A., Le Guillou F. et al. (1985): Permixon versus placebo. Résultats d’une étude multicentrique. C.R. Thérap. Pharmacol. Clin., 4, 16–21.
Descotes J.L., Rambeaud J.J., Deschaseaux P., Faure G. (1995): Placebo-controlled evaluation of the efficacy and tolerability of Permixon® in benign prostatic hyperplasia after exclusion of placebo responders. Clin. Drug Invest., 9, 291.
Dathe G., Schmid H. (1991): Phytotherapie der benignen Protatahyperplasie (BPH) mit ExtractumSerenoa repens (Permixon). Urologe [B], 31, 220–223.
Sommer R.G. (1991): Sabalextract hat hohe therapeutische Qualität. Therapiewoche, 43, 114–115.
Boccafoschi C., Annoscia S. (1983): Confronto fra estratto diSerenoa repens e placebo mediante prova clinica controlate in pazienti con adenomatosi prostatica. Urologia, 30, 1257–1269.
Emili E., Lo Cigno M., Petrone U. (1983): Risultati clinici su un nuovo farmaco nelle terapia dell’ipertrofia della prostata (Permixon). Urologia, 30, 1032–1049.
Mandressi A., Tarallo U., Maggioni A., Tombolini P., Rocco F., Quadraccia P. (1987): Treatment of uncomplicated benign prostatic hypertrophy (BPH) by an extract of Serenoa repens. Clinical results. J. Endocrinol. Invest., 10 (Suppl. 2), 49–55.
Tasca A., Barulli M., Cavazzana A., Zattoni F., Artibani W., Pagano F. (1985): Trattamento della sintomatologia ostruttiva da adenoma prostatico con estratto diSerenoa repens. Minerva Urol. Nefrol., 37, 87–91.
Olle Carreras J. (1987): Our experience with hexanic extract ofSerenoa repens in the treatment of benign prostatic hypertrophy. Arch. Espan. Urologia, 40, 310–313.
Neuzil E., Cousse H. (1993): Le palmier-scieSerenoa repens. Aspects pharmacologiques. Utilisation thérapeutique actuelle. Bull. Soc. Pharm. Bordeaux, 132, 142–163.
Breu W., Hagenlocher M., Redl K., Tittel G., Stadler F., Wagner H. (1992): Antiphlogistische Wirkung eines mit hyperkritischem Kohlendioxid gewonnenen Sabalfrucht-Extractes. In-vitro-Hemmung des Cyclooxygenase-und 5-Lipoxygenase Metabolismus. Arzneimittelforschung, 42, 547–551.
Tarayre J.P., Delhon A., Lauressergues H. et al. (1983): Action anti-oedémateuse d’un extrait hexanique de drupes deSerenoa repens Bartr. Ann. Pharm. Fr., 41, 559–570.
Carilla E., Briley M., Fauran F., Sultan C., Devillier C. (1984): Binding of Permixon, a new treatment for prostatic benign hyperplasia, to the cytosolic androgen receptor in the rat prostate. J. Steroid Chem., 20, 521–523.
Vacher P., Prevarskaya N., Skryma R. et al. (1995): The lipidosterolic extract fromSerenoa repens interferes with prolactin receptor signal transduction. J. Biomed. Sci., 2, 357–365.
Delos S., Iehle C., Martin P.M., Raynaud J.P. (1994): Inhibition of the activity of basic 5α-reductase (type 1) detected in DU 145 cells and expressed in insect cells. J. Steroid Chem., 48, 347–352.
Wajda-Dubos J.P., Farines M., Soulier J., Cousse H. (1996): Etude comparative de la fraction lipidique des pulpes et graines deSerenoa repens. Oléagineux Corps gras Lipides, 3, 136–139.
Lee Robinette C. (1988): Sex-hormone-induced inflammation and fibromuscular proliferation in the rat lateral prostate. The Prostate, 12, 271–286.
Ullberg S. (1954): Study on the distribution and fate of35S-labeled benzylpenicillin in the body. Acta Radiol., Suppl. 118, 1–110.
Benard P. (1991): Whole-body autoradiography of pharmaceuticals and chemicals. In: P.H. Bach & J.R.J. Baker, eds, Histochemical and immunohistochemical techniques: applications to pharmacology and toxicology. Chapman & Hall, London, pp 11–51.
Becker W., Bruce A. (1985): Autoradiographic studies with fatty acids and some other lipids: a review. Prog. Lipid Res., 24, 325–346.
Appelgren L.E. (1967): Sites of steroid hormone formation: autoradiographic studies using labelled precursors. Acta Physiol. Scand., Suppl. 301, 1–108.
Author information
Authors and Affiliations
Additional information
Footnote: Permixon® is marketed by Pierre Fabre Médicament (Castres, France) whose other trademarks include Capistan, Libeprosta and Sereprostat.
Rights and permissions
About this article
Cite this article
Chevalier, G., Benard, P., Cousse, H. et al. Distribution study of radioactivity in rats after oral administration of the lipido/sterolic extract ofSerenoa repens (Permixon®) supplemented with [1-14C]-lauric acid, [1-14C]-oleic acid or [4-14C]-β-sitosterol. European Journal of Drug Metabolism and Pharmacokinetics 22, 73–83 (1997). https://doi.org/10.1007/BF03189787
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03189787