Abstract
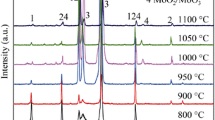
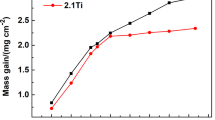
A Ni3Al-based alloy, the composition of which was Ni-16.0% Al-7.8% Cr-1.3% Zr-0.8% Mo-0.025%B, was cyclically oxidized in the temperature range of 900 to 1100°C in air for up to 500 hr. The alloy displayed good cyclic oxidation resistance up to 1000°C, with little scale spallation. It, however, lost cyclic oxidation resistance during oxidation at 1100°C after about 200 hr, displaying large weight losses due to serious scale spallation. NiO, α-Al2O3, NiAl2O4 and ZrO2 were formed. The oxide scales consisted primarily of an outer Ni-rich layer which was prone to spallation, and (Al, Cr, Zr, Mo, Ni)-containing internal oxides which were adherent due mainly to the formation of (Al2O3, ZrO2)-containing oxides that keyed the oxide scale to the matrix alloy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
C. T. Liu and K. S. Kumar,JOM 45, May, 38 (1993).
S. C. Choi, H. J. Cho, and D. B. Lee,Oxid. Met. 46, 109 (1996).
S. Taniguchi and T. Shibata,Oxidation of High-Temperature Intermetallics (eds., T. Grobstein and J. Doychak), p. 17, TMS, Warrendale, PA (1988).
J. Klöwer, U. Brill, and U. Heubner,Inermetallics 7, 1183 (1999).
T. T. Chuang, Y. C. Pan, and T. H. Chuang,J. Alloys & Compounds 243, 126 (1996).
Y. C. Pan, T. H. Chuang, and Y. D. Yao,J. Mater. Sci. 26, 6097 (1991).
W. W. Lee and D. B. Lee,J. Corros. Sci. Soc. of Kor. 27, 603 (1998).
I. Barin,Thermochemical Data of Pure Substances VCH, Weinhein, Germany (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, GY., Lee, DB. Cyclic oxidation behavior of Ni3Al-7.8% Cr-1.3% Zr-0.8% Mo-0.025% B between 900 and 1100°C. Met. Mater. Int. 8, 375–379 (2002). https://doi.org/10.1007/BF03186110
Issue Date:
DOI: https://doi.org/10.1007/BF03186110