Abstract
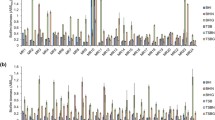
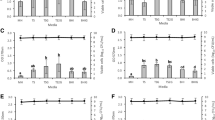
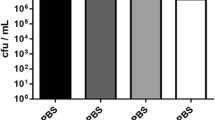
Biofilm production is an important step in the pathogenesis ofStaphylococcus epidermidis associated biomaterial infections.Staphylococcus epidermidis strains isolated from dialysis fluid (n=9) and needle cultures (n=14) were phenotyped and genotyped for extracellular polysaccharide production and were examined for their ability to produce slime in a medium at various pH levels (3, 5, 7, 9 and 12) and with ethanol supplementation (0, 2, 5, 10 and 15%) using a semi-quantitative adherence assay. A total of 23 clinicalicaADBC positiveS. epidermidis, one reference strain (S. epidermidis CIP 106510) used as positive control, and oneicaADBC negative strain (E21) were investigated. Qualitative biofilm production analysis revealed that 15 of the 23icaADBC positive strains (65.21%) produced slime on Congo Red agar plates. Quantitative biofilm was determined by measuring the optical density at 570 nm (OD570). The results show that the slime production depended on the pH value of the medium and the ethanol concentration. At highly acidic (pH 3) and alkaline (pH 12) levels, the OD570 was lower, while at pH 7 the adhesion was moderate. In addition the cells adhered strongly with 2% ethanol than with the other concentrations. Our results suggest that pH and ethanol were stress factors that led toS. epidermidis biofilm formation and also play a possible role in the pathogenesis of biomaterial-related infections.
Article PDF
Similar content being viewed by others
References
Arciola C.R., Campoccia D., Gamberini S., Cervellati M., Donati E., Montanaro L. (2002). Detection of slime production by means of an optimised Congo red agar plate test based on a colourimetric scale inStaphylococcus epidermidis clinical isolates genotyped forica locus. Biomaterials, 23: 4233–4239.
Bayer M.E., Sloyer J.L.J. (1990). The electrophoretic mobility of Gram-negative and Gram-positive bacteria: an electrokinetic analysis. J. Gen. Microbiol., 136: 867–874.
Cerca N., Pier G.B., Vilanova M., Oliveira R., Azeredo J. (2005). Quantitative analysis of adhesion and biofilm formation on hydrophilic and hydrophobic surfaces of clinical isolates ofStaphylococcus epidermidis. Res. Microbiol., 156: 506–514.
Christensen G.D., Simpson W.A., Younger J.J., Baddour L.M., Barrett F.F., Melton D.M., Beachey E.H. (1985). Adherence of coagulase-negative staphylococci to plastic tissue culture plates: a quantitative model for the adherence of staphylococci to medical devices. J. Clin. Microbiol., 22: 996–1006.
Conlon K.M., Humphreys H., O’Gara J.P. (2002).icaR encodes a transcriptional repressor involvedin environmental regulation ofica operon expression and biofilm formation inStaphylococcus epidermidis. J. Bacteriol., 184: 4400–4408.
Costerton J.W., Stewart P.S., Greenberg E.P. (1999). Bacterial biofilms: a common cause of persistent infections. Science, 284: 1318–1322.
Doyle R. (2000). Contribution of the hydrophobic effect to microbial infection, Microb. Infect. 2: 39–400.
Elliott T.S., Faroqui M.H., Armstrong R.F., Hanson G.C. (1994). Guidelines for good practice in central venous catheterization. J. Hosp. Infect., 28: 163–176.
Fitzpatrick F., Humphreys H., Smyth E., Kennedy C.A., O’Gara J.P. (2002). Environmental regulation of biofilm formation in intensive care unit isolates ofStaphylococcus epidermidis. J. Hosp. Infect., 42: 212–218.
Freeman D.J., Falkiner F.R., Keane C.T. (1989). New method for detecting slime production by coagulase-negative staphylococci. J. Clin. Pathol., 42: 872–874.
Götz F. (2002).Staphylococcus and biofilms. Mol. Microbiol., 43: 1367–1378.
Knobloch J.K., Bartscht K., Sabottke A., Rohde H., Feucht H.H., Mack D. (2001). Biofilm formation byStaphylococcus epidermidis depends on functionalRsbU, an activator of thesigB operon: differential activation mechanisms due to ethanol and salt stress. J. Bacteriol., 183: 2624–2633.
Lawrence E.L., Turner I.G. (2005). Materials for urinary catheters: a review of their history and development in the UK. Med. Eng. Phys., 27: 443–453.
Mack D., Bartscht K., Fischer C., Rohde H., de Grahl C., Dobinsky S., Horstkotte M.A., Kiel K., Knobloch J.K. (2001). Genetic and biochemical analysis ofStaphylococcus epidermidis biofilm accumulation. Method. Enzymol., 336: 215–239.
Mack D., Rohde H., Dobinsky S., Riedewald J., Nedelmann M., Knobloch J.K., Elsner H.A., Feucht H.H. (2000). Identification of three essential regulatory gene loci governing expression of theStaphylococcus epidermidis polysaccharide intercellular adhesin and biofilm formation. Infect. Immun., 68: 3799–807.
McKenney D., Pouliot K.L., Wang Y., Murthy V., Ulrich M., Döring G., Lee J.C., Goldmann D.A., Pier G.B. (1999). Broadly protective vaccine forStaphylococcus aureus based on anin vivo expressed antigen. Science, 284: 1523–1527.
Meinders J.M., van der Mei H.C., Busscher H.J. (1994). Physicochemical aspects of deposition ofStreptococcus thermophilus B to hydrophobic and hydrophilic substrata in a parallel plate flow chamber. J. Colloid. Interf. Sci., 164: 355–363.
Memple M., Schmidt T., Weidinger S., Weidinger S., Schnopp Ch., Foster T., Ring J., Abeck D. (1998). Role ofStaphylococcus aureus surface-associated proteins in the attachment to cultured HaCa T keratinocytes in a new adhesion assay. J. Invest. Dermatol., 111: 452–456.
Mermel L.A. (2000). Prevention of intravascular catheter-related infections. Ann. Intern. Med., 132: 391–402.
Patrick C.C., Plaunt M.R., Hetherington S.V., May S.M. (1992). Role ofStaphylococcus epidermidis slime layer in experimental tunnel tract infections. Infect. Immun., 60: 1363.
Pearson M.L. (1996). Guideline for prevention of intravascular device-related infections. Hospital Infection Control Practices Advisory Committee. Infect. Cont. Hosp. Ep., 17: 438–473.
Peters G., Locci R., Pulverer G. (1982). Adherence and growth of coagulase-negative staphylococci on surfaces of intravenous catheters. J. Infect. Dis., 146: 479–482.
Rachid S., Ohlsen K., Witte W., Hacker J., Ziebuhr W. (2000a). Effect of subinhibitory antibiotic concentrations on polysaccharide intercellular adhesin expression in biofilm-formingStaphylococcus epidermidis. Antimicrob. Agents Chemother., 44: 3357–3363.
Rachid S., Cho S., Ohlsen K., Hacker J., Ziebuhr W. (2000b). Induction ofStaphylococus epidermidis biofilm formation by environmental factors: the possible involvement of the alternative transcription factor SigB.In L. Emody, G. Blum-Oehler, J. Hacker, Pal T., Eds, Genes and Proteins Underlying Microbial Urinary Tract Virulence. Plenum Press, New York, N.Y., pp. 159–166.
Rupp M.E., Archer G.L. (1994). Coagulase-negative staphylococci: pathogens associated with medical progress. Clin. Infect. Dis., 19: 231–243.
Shapiro J.A. (1998). Thinking about bacterial populations as multicellular organisms. Annu. Rev. Microbiol., 52: 81–104.
Vasudevan P., Nair M.K., Annamalai T., Venkitanarayanan K.S. (2003). Phenotypic and genotypic characterization of bovine mastitis isolates ofStaphylococcus aureus for biofilm formation. Vet. Microbiol., 92: 179–185.
Von Eiff C., Heilmann C., Peters G. (1999). New aspects in the molecular basis of polymer-associated infections due to staphylococci. Eur. J. Clin. Microbiol. Infect. Dis., 18: 843–846.
White A., Handler P., Smith E.L. (1978). Enzymes I, nature, classification, kinetics, metabolic inhibitors: control of enzymatic activity. In: White A., Ed., Principles of Biochemistry, McGraw-Hill, Tokyo, pp. 196–230.
Zmantar T., Chaieb K., Miladi H., Mahdouani K., Bakhrouf A. (2006). Detection of the intercellular adhesion loci (ica) in clinicalStaphylococcus aureus strains responsible for hospital acquired auricular infection. Ann. Microbiol., 56: 349–352
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaieb, K., Chehab, O., Zmantar, T. et al. In vitro effect of pH and ethanol on biofilm formation by clinicalica-positiveStaphylococcus epidermidis strains. Ann. Microbiol. 57, 431–437 (2007). https://doi.org/10.1007/BF03175085
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175085