Abstract
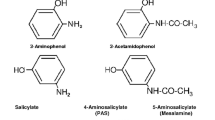
Ascorbate (AH-) and certain other biological reductants have long been known to produce the cytotoxic hydroxyl radical (OH) when oxidized by hydrogen peroxide (H2O2) in the presence of copper or iron catalysts. The present study documents the in vitro production of the OH solely from the oxidation of AH- by H2O2, independent of mediation by transition metals. Hydroxyl radical generation resulting from the AH-/H2O2 system was quantitatively documented by the specific radical-mediated hydroxylation of salicylic acid, a reaction that was readily assayed with HPLC coupled with electrochemical detection. Two ascorbate-copper complexes (e.g., AH-/Cu2+-EDTA/H2O2 and AH-/Cu2+-EDTA) and a copper/H2O2 system also generated OH, but less effectively than the AH-/H2O2 system. The ability of AH- and H2O2 to generate cytotoxic OH documents a reaction mechanism that may account for cytotoxic activity in some cellular environments where metal catalysts are lacking.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andersson, M. and Grankvist, K. (1995). Ascorbate-induced free radical toxicity to isolated islet cells. International Journal of Biochemistry & Cell Biology27, 493–498.
Andorn, A. C, Britton, R. S. and Bacon, B. R. (1996). Ascorbate-stimulated lipid peroxidation in human brain is dependent on iron but not on hydroxyl radical. Journal of Neurochemistry67, 717–722.
Arakawa, N., Nemoto, S., Suzuki, E. and Otsuka, M. (1994). Role of hydrogen peroxide in the inhibitory effect of ascorbate on cell growth. J Nutr Sci Vitaminol (Tokyo)40, 219–27.
Aruoma, O.I., Grootveld, M. and Halliwell, B. (1987). The Role of Iron in Ascorbate-Dependent Deoxyribose Degradation Evidence Consistent With a Site-Specific Hydroxyl Radical Generation Caused By Iron Ions Bound to the Deoxyribose Molecule. Journal of Inorganic Biochemistry29, 289–300.
Aruoma, O. I., Halliwell, B., Gajewski, E. and Dizdaroglu, M. (1991). Copper Ion-Dependent Damage to the Bases in Dna in the Presence of Hydrogen Peroxide. Biochemical Journal273, 601–604.
Asada, K. (1996). Radical production and scavenging in the chloroplasts. In Photosynthesis and the Environment (ed. N.R. Baker), pp. 123–150. Dordrecht, Kluwer., The Netherlands.
Banhegyi, G., Braun, L., Csala, M., Puskas, F. and Mandl, J. (1997). Ascorbate metabolism and its regulation in animals. Free Radic Biol Med23, 793–803.
Biaglow, J. E., Manevich, Y., Uckun, F. and Held, K. D. (1997). Quantitation of hydroxyl radicals produced by radiation and copper-linked oxidation of ascorbate by 2-deoxy-D-ribose method. Free Radic Biol Med22, 1129–38.
Chakrabarti, S., Mahmood, A., Kassis, A. I., Bump, E. A., Jones, A. G. and Makrigiorgos, G. M. (1996). Generation of hydroxyl radicals by nucleohistone-bound metal-adriamycin complexes. Free Radic Res25, 207–20.
Chin, H.-W. and Lindsay Robert, C. (1994). Ascorbate and transition-metal mediation of methanethiol oxidation to dimethyl disulfide and dimethyl trisulfide. Food Chemistry49, 387–392.
Chiueh, C. C, Krishna, G., Tulsi, P., Obata, T., Lang, K., Huang, S.-J. and Murphy, D. L. (1992). Intracranial microanalysis of salicylic acid to detect hydroxyl radical generation through dopamine autooxidation in the caudate nucleus: effects of MPP+. Free Rad. Biol. Med.13, 581–583.
Chiueh, C. C, Murphy, D. L., Lang, M. K., Tulsi, P. K. and Huang, S.-J. (1993). Hydroxyl free radical (OH) formation reflected by salicylate hydroxylation and neuromelanin:In vivo markers for oxidant injury of nigral neurons. Ann. New York Acad. Sci.679, 370–375.
Cohen, G. (1984). Oxy-radical toxicity in catecholamine neurons. Neurotoxicology5, 77–82.
De Laurenzi, V., Melino, G., Savini, I., Annicchiarico-Petruzzelli, M., Finazzi-Agro, A. and Avigliano, L. (1995). Cell death by oxidative stress and ascorbic acid regeneration in human neuroectodermal cell lines. Eur J Cancer31A, 463–6.
Dizdaroglu, M., Rao, G., Halliwell, B. and Gajewski, E. (1991). Damage to the DNA bases in mammalian chromatin by hydrogen peroxide in the presence of ferric and cupric Ions. Archives of Biochemistry & Biophysics285, 317–324.
Drouin, R., Rodriguez, H., Gao, S. W., Gebreyes, Z., TR, O. C, Holmquist, G. P. and Akman, S. A. (1996). Cupric ion/ascorbate/hydrogen peroxide-induced DNA damage: DNA-bound copper ion primarily induces base modifications. Free Radic Biol Med21, 261–73.
Elzanowska, H., Wolcott, R. G., Hannum, D. M. and Hurst, J. K. (1995). Bactericidal properties of hydrogen peroxide and copper or iron-containing complex ions in relation to leukocyte function. Free Radic Biol Med18, 437–49.
Florence, T. M. (1984). The Production of Hydroxyl Radical From Hydrogen Peroxide. Journal of Inorganic Biochemistry22, 221–230.
Grinberg, L. N., Rachmilewitz, E. A., Kitrossky, N. and Chevion, M. (1995). Hydroxyl radical generation in beta-thalassemic red blood cells. Free Radical Biology & Medicine18, 611–615.
Halliwell, B. and Gutteridge, J. M. C. (1999). Free Radicals in Biology and Medicine, 3 edition. Oxford University Press, Oxford.
Hermes-Lima, M., Wang, E. M., Schulman, H. M., Storey, K. B. and Ponka, P. (1994). Deoxyribose degradation catalyzed by Fe(III)-EDTA: Kinetic aspects and potential usefulness for submicromolar iron measurements. Molecular & Cellular Biochemistry137, 65–73.
Hisanga, K., Sagar, M.D. and Sharp, F. R. (1992). Ascorbate neurotoxicity in cortical cell cultures. Ann. Neurol.31, 562–565.
Juven, B. J. and Kanner, J. (1986). Effect of Ascorbic Isoascorbic and Dehydroascorbic Acids On the Growth and Survival of Campylobacter-Jejuni. Journal of Applied Bacteriology61, 339–346.
Kaur, H., Whiteman, M. and Halliwell, B. (1997). Peroxynitrite-dependent aromatic hydroxylation and nitration of salicylate and phenylalanine. Is hydroxyl radical involved? Free Radic Res26, 71–82.
Makrigiorgos, G. M., Bump, E., Huang, C, Baranowska-Kortylewicz, J. and Kassis, A. I. (1995). A fluorimetric method for the detection of copper-mediated hydroxyl free radicals in the immediate proximity of DNA. Free Radic Biol Med18, 669–78.
Miura, T., Muraoka, S. and Ogiso, T. (1994). Effect of ascorbate on adriamycin-Fe-3+-induced lipid peroxidation and DNA damage. Pharmacology & Toxicology74, 89–94.
Miwa, N., Yamazaki, H., Nagaoka, Y., Kageyama, K., Onoyama, Y., Matsui Yuasa, I., Otani, S. and Morisawa, S. (1988). Altered Production of the Active Oxygen Species Is Involved in Enhanced Cytotoxic Action of Acylated Derivatives of Ascorbate to Tumor Cells. Biochimica et Biophysica Acta972, 144–151.
Murata, A., Oshima, K., Kubo, T., Yokoo, K., Tanaka, A. and Kato, F. (1985). Mechanism of Synergistic Effect of Ascorbic-Acid and Vitamin K-5 in Inactivation of Phage J-l. Vitamins59, 203–210.
Murata, A., Suenaga, H., Hideshima, S., Tanaka, Y. and Kato, F. (1986). Hydroxyl Radical As the Reactive Species in the Inactivation of Phages By Ascorbic-Acid. Agricultural & Biological Chemistry50, 1481–1488.
Nappi, A. J. and Vass, E. (1996). Hydrogen peroxide generation associated with the oxidations of the eumelanin precursors 5,6-dihydroxyindole and 5.6-dihydroxyindole-2-carboxylic acid. Melanoma Research6, 341–349.
Nappi, A. J. and Vass, E. (1997). Comparative studies of enhanced iron-mediated production of hydroxyl radical by glutathione, cysteine, ascorbic acid and selected catechols. Biochim Biophys Acta1336, 295–302.
Nappi, A. J. and Vass, E. (1998a). Hydroxyl radical formation resulting from the interaction of nitric oxide and hydrogen peroxide. Biochim. Biophys. Acta1380, 55–63.
Nappi, A. J. and Vass, E. (1998b). Hydroxyl radical formation via iron-mediated Fenton chemistry is inhibited by methylated catechols. Biochim. Biophys. Acta 1425), 159-167.
Nappi, A. J., Vass, E. and Collins, M. A. (1999). Contrasting effects of catecholic and O-methylated tetrahydroisoquinolines on hydroxyl radical production. Biochim. Biophys. Acta.1434, 64–73.
Nappi, A. J., Vass, E., Prota, G. and Memoli, S. (1995). The effects of hydroxyl radical attack on dopa, dopamine, 6-hydroxydopa and 6-hydroxydopamine. Pigment Cell Res.8, 283–293.
Nemoto, S., Otsuka, M. and Arakawa, N. (1996a). Inhibitory effect of ascorbate on cell growth: relation to catalase activity. J Nutr Sci Vitaminol (Tokyo)42, 77–85.
Nemoto, S., Otsuka, M. and Arakawa, N. (1996b). A positive correlation between catalase activity and ascorbate uptake in the tissues of guinea pigs and cultured cells of mammals. J Nutr Sci Vitaminol (Tokyo)42, 387–95.
Niki, E., Noguchi, N., Tsuchihashi, H. and Gotoh, N. (1995). Interaction among vitamin C, vitamin E and beta-carotene. American Journal of Clinical Nutrition 62.
Nowak, D., Piasecka, G., Antczak, A. and Pietras, T. (1991). Effect of Ascorbic Acid On Hydroxyl Radical Generation By Chemical Enzymatic and Cellular Systems Importance For Antioxidant Prevention of Pulmonary Emphysema. Biomedica Biochimica Acta50, 265–272.
Schmidt, R. J., Chung, L. Y., Andrews, A. M. and Turner, T. D. (1993). Toxicity of L-ascorbic acid to L929 fibroblast cultures: relevance to biocompatibility testing of materials for use in wound management. J Biomed Mater Res27, 521–30.
Sestili, P., Brandi, G., Brambilla, L., Cattabeni, F. and Cantoni, O. (1996). Hydrogen peroxide mediates the killing of U937 tumor cells elicited by pharmacologically attainable concentrations of ascorbic acid: cell death prevention by extracellular catalase or catalase from cocultured erythrocytes or fibroblasts. J Pharmacol Exp Ther277, 1719–25.
Tajima, M., Toguchi, M., Kanda, Y., Kunii, S., Hosaka, M., Arakawa, H., Maeda, M., Satoh, K., Asano, K., Kochi, M. and Sakagami, H. (1998). Role of hydrogen peroxide for cell death induction by sodium 5,6-benzylidene-L-ascorbate. Anticancer Research18, 1697–1702.
Ueda, J.-I., Saito, N., Shimazu, Y. and Ozawa, T. (1996). A comparison of scavenging abilities of antioxidants against hydroxyl radicals. Archives of Biochemistry & Biophysics333, 377–384.
Willekens, H., Chamnongpol, S., Davey, M., Schraudner, M., Langebartels, C, Van Montagu, M., Inze, D. and Van Camp, W. (1997). Catalase is a sink for H202 and is indispensable for stress defence in C3 plants. Embo J16, 4806–16.
Woo, K. I. and Lee, J. (1995). The effects of ascorbic acid on free radical injury in cultured retinal pigment epithelial cells. Korean J Ophthalmol9, 19–25.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nappi, A.J., Vass, E. Hydroxyl radical production by ascorbate and hydrogen peroxide. neurotox res 2, 343–355 (2000). https://doi.org/10.1007/BF03033342
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF03033342