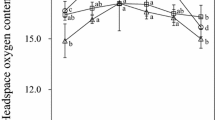
Abstract
Fatty acid esters, high in linoleic acid, were prepared and stored for long-term engine tests. Storage tests with these esters were undertaken to obtain more information on optimal storage requirements and general stability characteristics.
Samples were kept at three temperature levels (20 C, 30 C and fluctuating around 50 C) for a 90-day period and were removed at regular intervals for chemical and physical analysis. The influence of air, temperature, light, TBHQ and contact with mild steel was evaluated by comparing the free fatty acid, peroxide, anisidine, ultraviolet absorption, viscosity and induction periods. A statistical model was used to evaluate the data and to reduce the large number of data points to comparable curves.
Storage of esters in contact with air, especially at a temperature above 30 C, resulted in significant increases in peroxide, ultraviolet absorption, free fatty acid, viscosity and anisidine values. Exclusion of air retarded oxidation at all temperature levels.
A direct relationship between viscosity increases and oxidation parameters was evident. Exposure to light caused a small increase in the oxidation parameters of esters stored at the highest temperature level. Addition of TBHQ prevented oxidation of samples stored under moderate conditions. Under unfavorable storage conditions the anti-oxidant was no longer effective. Mild steel had very little effect on the oxidation parameters. Only the anisidine values of samples stored at the highest temperature level were slightly increased. Methyl esters performed slightly better than ethyl esters during the storage test.
The following practical guidelines for storage of fatty acid ester fuels are: (i) airtight containers should be used; (ii) the storage temperature should be <30 C; (iii) mild steel (rust free) containers may be used, and (iv) TBHQ has a beneficial effect on oxidation stability.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Hawkins, C.S., and J. Fuls, Proceedings of the International Conference on Plant and Vegetable Oils as Fuels. American Society of Agricultural Engineers, St. Joseph, MI, 1982, p. 184.
Du Plessis, L.M., F.J.C. Hugo and A. Engelbrecht, Internal combustion engine fuel. South African Patent Application no. 81/7532, 1981.
Fuls, J., and F.J.C. Hugo, World overview of plant oils for fuels: Status of research and applications technology. Third International Conference on Energy Use Management, West Berlin, Oct. 26–30, 1981, Pergamon Press, p. 1595–1602.
Kusy, P.F., Proceedings of the International Conference on Plant and Vegetable Oils as Fuels, American Society of Agricultural Engineers, St. Joseph, MI, 1982, p. 127.
Romano, S. Ibid. p. 106.
Du Plessis, L.M., Report to Evaluation Meeting, Energy in Transportation, National Programme for Energy Research, CSIR, Pretoria, Sept. 9, 1982.
Du Plessis, L.M., and J.B.M. de Villiers, Vegetable oil as diesel fuel, Seminar III, USDA Northern Regional Research Center, Peoria, IL, Oct. 19–20, 1983.
Dittmer, J.C., and R.L. Lester, J. Lipid Res. 5:126 (1964).
International Organization for Standardization TC 34, ISO 660, 1968.
Ibid. TC 34, ISO 3960, 1977.
Ibid. TC 34, ISO 6885, 1981.
Hadorn, H., In: Functional Properties of Fats in Foods, J. Solms, ed., Forster Publishing Ltd., Zürich, 1973, p. 81.
Aitken, M.A., Technometrics, 16:221 (1974).
Author information
Authors and Affiliations
About this article
Cite this article
Du Plessis, L.M., De Villiers, J.B.M. & Van Der Walt, W.H. Stability studies on methyl and ethyl fatty acid esters of sunflowerseed oil. J Am Oil Chem Soc 62, 748–752 (1985). https://doi.org/10.1007/BF03028746
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03028746