Abstract
Purpose
To determine whether low doses of droperidol mixed with morphine in patient-controlled analgesia (PCA) would extend the duration of prophylaxis against postoperative nausea and vomiting.
Methods
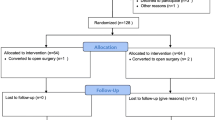
Healthy women having elective open-abdominal gynaecological surgery consented to this double-blind, place-bo-controlled study. Subjects were randomized to receive placebo, or 1 mg droperidol before induction followed by droperidol 0.0 (bolus group), 0.02 (0.02 group), or 0.04 (0.04 group) mg · mg−1 of PCA morphine. Study endpoints included severity of nausea, episodes of vomiting and rescue antiemetic doses, pain, and sedation and were assessed at 1, 2, 4, 8, 12, 16, 20 and 24 hr postoperatively.
Results
Seventy-one subjects completed the study. The groups were similar in age, weight, surgical time, pain scores, and morphine used. The 0.04 group had lower mean visual analogue scale scores for nausea (P < 0.05 vs all other groups). The incidence of vomiting was lower in all treatment groups (P < 0.05 for all groups vs placebo). The 0.04 group had lower rescue antiemetic requirements than the bolus group (P < 0.03). Mean sedation scores were tow in all groups but were increased with PCA droperidol (P < 0.02).
Conclusions
Droperidol 1 mg before induction of anaesthesia reduces postoperative vomiting. The addition of droperidol 0.04 mg · mg−1 of PCA morphine further reduces (i) severity of nausea and (ii) rescue antiemetic requirements postoperatively. No clinically significant side-effects were attributed to this regimen.
Résumé
Objectif
Établir si de faibles doses de dropéridol combinées à la morphine pour l’analgésie auto-contrôlée (PCA) pouvent prolonger la prévention antiémétique.
Méthodes
Des femmes en bonne santé soumises à une chirurgie gynécologique abdominale ouverte ont consenti à participer à cette étude en double aveugle et contrôlée avec placebo. Les patientes ont été réparties aléatoirement pour recevoir soit un placebo, soit dropéridol 1 mg avant l’induction suivi de dropéridol 0,0 (groupe bolus), 0,02 (groupe 0,02) ou 0,04 (groupe 0,04) mg · mg−1 de morphine en PCA. Les données recueillies portaient sur la gravité des nausées, les épisodes de vomissements et les doses d’antiémétiques de sauvetage, sur la douleur et la sédation, après 1, 2, 4, 8, 12, 16, 20 et 24 de l’intervention.
Résultats
Soixante et onze patientes ont complété l’étude. Les groupes étaient identiques quant à l’âge, le poids, la durée de la chirurgie, les scores de douleur et la morphine administrée. Le groupe 0,04 présentait les scores moyens les plus bas sur l’échelle visuelle analogique pour la nausée (P < 0,05) vs tous les autres groupes). L’incidence des vomissements était inférieure pour tous les groupe traités (P < 0,05 pour tous les groupes vs placebo). Le groupe 0,04 avait des besoins moins importants d’antiémétiques de sauvetage que le groupe bolus (P < 0,03). Les scores moyens de sédation étaient bas dans tous les groupes mais augmentaient avec le dropéridol en PCA (P < 0,02).
Conclusion
Le dropéridol 1 mg avant l’induction diminue les vomissements postopératoires. L’addition de dropéridol 0,04 mg · mg−1 à la morphine en PCA diminue encore plus (i) la gravité des nausées et (ii) les besoins d’antiémétiques de sauvetage postopératoires. Aucun effet secondaire important n ’a été noté.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kenny GNC. Risk factors for postoperative nausea and vomiting. Anaesthesia 1994; 49: S6–10.
White PF, Shafer A. Nausea and vomiting: causes and prophylaxis. Semin Anesth 1987; 6: 300–8.
Watcha MF, White PF. Postoperative nausea and vomiting. Its etiology, treatment, and prevention. Anesthesiology 1992; 77: 162–84.
Pataky AO, Ktiz DS, Andrews RW, Lecky JH. Nausea and vomiting following ambulatory surgery: are all procedures created equal? Anesth Analg 1988; 67: S163.
Vance JP, Neill RS, Norris W. The incidence and aetiology of post-operative nausea and vomiting in a plastic surgery unit. Br J Plastic Surg 1973; 26: 336–9.
Patton CM Jr, Moon MR, Dannemiller FJ. The prophylactic antiemetic effect of droperidol. Anesth Analg 1974; 53: 361–4.
Egbert AM, Parks LH, Short LM, Burnett ML. Randomized trial of postoperative patient-controlled analgesia vs intramuscular narcotics in frail elderly men. Arch Intern Med 1990; 150: 1897–903.
White PF. Use of patient-controlled analgesia for the management of acute pain. JAMA 1988; 259: 243–7.
Williams OA, Clarke FL, Harris RW, Smith P, Peacock JE. Addition of droperidol to patient-controlled analgesia: effect on nausea and vomiting. Anaesthesia 1993; 48: 881–4.
Sharma SK, Davies MW. Patient-controlled analgesia with a mixture of morphine and droperidol. Br J Anaesth 1993; 71: 435–6.
Barrow PM, Hughes DG, Redfern N, Urie J. Influence of droperidol on nausea and vomiting during patient-controlled analgesia. Br J Anaesth 1994; 72: 460–1.
Gan TJ, Alexander R, Fennelly M, Rubin AP. Comparison of different methods of administering droperidol in patient-controlled analgesia in the prevention of postoperative nausea and vomiting. Anesth Analg 1995; 80: 81–5.
McKenzie R, Tantisira B, Jackson D, Bach T, Riley T. Antiemetic efficacy of a droperidol-morphine combination in patient-controlled analgesia. J Clin Anesth 1995; 7: 141–7.
Melnick B, Sawyer R, Karambelkar D, Phitayakorn P, Lim Uy NT, Patel R. Delayed side effects of droperidol after ambulatory general anesthesia. Anesth Analg 1989; 69: 748–51.
Melnick BM. Extrapyramidal reactions to low-dose droperidol. Anesthesiology 1988; 69: 424–6.
Dupre LJ, Stieglitz P. Extrapyramidal syndromes after premedication with droperidol in children. Br J Anaesth 1980; 52: 831–3.
Rowbotham DJ. Current management of postoperative nausea and vomiting. Br J Anaesth 1992; 69: S46–59.
O’Donovan N, Shaw J. Nausea and vomiting in day-case dental anaesthesia. The use of low-dose droperidol. Anaesthesia 1984; 39: 1172–6.
Mortensen PT. Droperidol (Dehydrobenzperidol®): postoperative anti-emetic effect when given intravenously to gynaecological patients. Acta Anaesthesiol Scand 1982; 26: 48–52.
van den Berg AA, Lambourne A, Yazji NS, Laghari NA. Vomiting after ophthalmic surgery. Effects of intra-operative antiemetics and postoperative oral fluid restriction. Anaesthesia 1987; 42: 270–6.
Cressman WA, Plostnieks J, Johnson PC. Absorption, metabolism and excretion of droperidol by human subjects following intramuscular and intravenous administration. Anesthesiology 1973; 38: 363–9.
Harris AL. Cytotoxic-therapy-induced vomiting is mediated via enkephalin pathways. Lancet 1982; 1: 714–6.
Stoelting RK. Pharmacology and Physiology in Anesthetic Practice, 2nd ed. Philadelphia: J.B. Lippincott Company, 1991: 70–101.
Willan AR, Warr D, Pater JL, Levitt M, Erlichman C, Osoba D. Methodological issues in antiemetic studies.In: Osoba D (Ed.). Effect of Cancer on Quality of Life. Boca Ratan: CRC Press, Inc., 1991: 229–49.
McCollum JSC, Milligan KR, Dundee JW. The antiemetic effect of propofol. Anaesthesia 1988; 43: 239–40.
Gunawardene RD, White DC. Propofol and emesis. Anaesthesia 1988; 43: S65–7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klahsen, A.J., O’Reilly, D., McBride, J. et al. Reduction of post-operative nausea and vomiting with the combination of morphine and dropéridol in patient-controlled analgesia. Can J Anaesth 43, 1100–1107 (1996). https://doi.org/10.1007/BF03011835
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03011835