Abstract
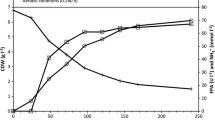
Among 18 tested strains ofClaviceps spp., 7 produced significant amounts of exocellular polysaccharide (EPS). The maximum production of EPS was found in fermentation broth ofClaviceps viridis. The kinetics of growth, substrate consumption, and EPS production in the batch, aerobic, submerged culture of this fungus were investigated in detail. The experimental data were processed by a simple mathematical model describing mass balance of growth, substrate consumption, formation of intermediates, and production of EPS. The parameters of the model were estimated from data obtained in cultivation performed in flasks and two laboratory fermentors of different size. Physiological similarity was obtained during process scale-up in volumetric ratio 1 : 100. The sugar consumption efficiency (52 %) and observed EPS productivity (1.9 kg/m3 per d) were comparable with literature data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- EPS:
-
exocellular polysaccharide
- k 1,k 2 :
-
rate constants in Eqs1–2, Lg−1d−1
- k 3,k 4 :
-
rate constants in Eqs1–4, 1/d
- K 1,K 2 :
-
reciprocal saturation constants in Eqs1–2, L/g
- K S :
-
saturation constant for sucrose in Eqs2–3, g/L
- μ:
-
specific growth rate, 1/d
- P:
-
polysaccharide concentration, g/L
- r P max :
-
maximum specific rate of EPS formation, 1/d
- S1 :
-
total concentration of monosaccharides, g/L
- S2 :
-
sucrose concentration, g/L
- t :
-
cultivation time, d
- T :
-
characteristic time for achievement of maximum EPS or dry biomass concentration, d
- VVM:
-
aeration rate, vessel volume per min
- X:
-
dry biomass concentration in fermentor, g/L
- Y:
-
macroscopic yield coefficient, defined by Eq.2
References
Banks G.T., Mantle P.G., Szczyrbak C.A.: Large-scale production of clavine alkaloids byClaviceps fusiformis.J.Gen.Microbiol. 82, 345–361 (1974).
Borošková Z., Reiterová K., Dubinský P., Tomašovicová O., Machnická B.: Inhibition of lymphoproliferative response and its restoration with a glucan immunomodulator in mice with experimental larval toxocarosis.Folia Microbiol. 43, 475–476 (1998).
Buck K.W., Chen A.W., Dickerson A.G., Chain E.H.: Formation and structure of extracellular glucans produced byClaviceps speciesJ.Gen.Microbiol. 51, 337–352 (1968).
Charalampopoulos D., Wang R., Pandiella S.S., Webb C.: Application of cereals and cereal components in functional foods: a review.Internat.J.Food Microbiol. 79, 131–141 (2002).
Dean T.R.:Cellular Degradative Processes. J. Wiley & Sons, New York 1978.
Flieger M., Votruba J., Křen V., Pažoutová S., Rylko V., Sajdl P., Reháček Z.: Physiological control and process kinetics of clavine alkaloid production byClaviceps purpurea 129 and 129/35.Appl.Microbiol.Biotechnol. 29, 181–185 (1988).
Flieger M., Linhartová R., Sedmera P., Zima J., Sajdl P., Stuchlik J., Cvak L.: New alkaloids ofClaviceps paspali.J.Nat.Prod. 52, 1003–1008 (1989).
Flieger M., Sedmera P., Havlíček V., Cvak L., Stuchlík J.: 10-OH-cis- and 10-OH-trans-paspalic acid amide — new alkaloids fromClaviceps paspali.J.Nat.Prod. 56, 810–814 (1993).
Flieger M., Wurst M., Shelby R.: Ergot alkaloids: sources, structure and analytical methods.Folia Microbiol. 42, 3–30 (1997).
Flieger M., Kantorova M., Prell A., Řezanka T., Votruba J.: Biodegradable plastics from renewable sources.Folia Microbiol. 48, 27–44 (2003a).
Flieger M., Kantorova M., Benada O., Kofroňová O., Sobotka M., Cvak J., Votruba J.: Preparation of nanoparticles by electrocoagulation from soluble exopolysaccharides produced byClaviceps viridis. Biotechnol.Bioeng., in press (2003b).
Van Geel-Schutten G.H., Flesch F., Ten Brink B., Smith M.R., Dijkhuizen L.: Screening and characterization ofLactobacillus strains producing large amounts of exopolysaccharides.Appl.Microbiol.Biotechnol. 50, 697–703 (1998).
Gutierrez A., Prieto A., Martinez A.T.: Structural characterization of extracellular polysaccharides produces by fungi of genusPleurotus.Carbohydr.Res. 281, 143–154 (1996).
Kantorová M., Kolinská R., Pažoutová S., Honzátko A., Havlíček V., Fijeger M.: Ergot alkaloids produced by submerged culture ofClaviceps zizaniae.J.Nat.Prod. 65, 1039–1040 (2002).
Koniček J., Lasík J., Šafář H.: Production of exopolysaccharide by a new mutant strain ofAchromobacter delicatulus using whey fermentation.Folia Microbiol. 38, 68–70 (1993).
Mucksova J., Babiček K., Pospišil M.: Particulate 1,3-β-d-glucan, carboxymethylglucan and sulfoethylglucan influence of their oral or intraperitoneal administration on immunological respondence of mice.Folia Microbiol. 46, 559–564 (2001).
Osaku C.A., Sassaki G.L., Zancan G.T., Iacomini M.: Studies on neutral exopolysaccharides produced by the ectomycorrhizaThelephora terrestris.FEMS Microbiol.Lett. 216, 145–149 (2002).
Pažoutová S.: The phylogeny and evolution of the genusClaviceps.Mycol.Res. 105, 275–283 (2001).
Pažoutová S., Flieger M., Sajdl P., Řehaček Z., Taisinger J., Bass A.: The relationship between intensity of oxidative metabolism and predominance of agroclavine or elymoclavine in submergedClaviceps purpurea cultures.J.Nat.Prod. 44, 225–236 (1981).
Schilling B.M., Rau U., Maier T., Fankhauser P.: Modeling and scale-up of the unsterile scleroglucan production process withSclerotium rolfsii ATCC 15205.Bioproc.Eng. 20, 195–201 (1999).
Selbmann L., Onofri S., Fenice M., Federici F., Petruccioli M.: Production and structural characterization of the exopolysaccharide of the Antarctic fungusPhoma herbarum CCFEE 5080.Res Microbiol. 153, 585–592 (2002).
Steinbuchel A., de Baets S., van Damme E.J. (Eds):Biopolymers, Vol. 6. Polysaccharides II: Polysaccharides from Eukaryotes. Wiley-VCH, Weinheim (Germany) 2002.
Taber W.A.: Sequential formation and accumulation of primary and secondary shunt metabolic products inClaviceps purpurea.Appl.Microbiol. 12, 321–326 (1964).
Volesky J., Votruba J.,Modeling and Optimization of Fermentation Processes. Elsevier Science Publishers, Amsterdam 1992.
Wasser S.P.: Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides.Appl.Microbiol.Biotechol. 60, 258–274 (2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Flieger, M., Kantorová, M., Benešová, T. et al. Kinetics of soluble glucan production byClaviceps viridis . Folia Microbiol 48, 633–638 (2003). https://doi.org/10.1007/BF02993470
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02993470