Abstract
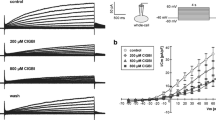
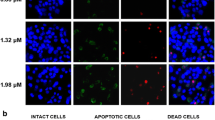
The effect of diazoxide, a K+ channel opener, on apoptotic cell death was investigated in HepG2 human hepatoblastoma cells. Diazoxide induced apoptosis in a dose-dependent manner and this was evaluated by flow cytometric assays of annexin-V binding and hypodiploid nuclei stained with propidium iodide. Diazoxide did not alter intracellular K+ concentration, and various inhibitors of K+ channels had no influence on the diazoxide-induced apoptosis; this implies that K+ channels activated by diazoxide may be absent in the HepG2 cells. However, diazoxide induced a rapid and sustained increase in intracellular Ca2+ concentration, and this was completely inhibited by the extracellular Ca2+ chelation with EGTA, but not by blockers of intracellular Ca2+ release (dantrolene and TMB-8). This result indicated that the diazoxideinduced increase of intracellular Ca2+ might be due to the activation of a Ca2+ influx pathway. Diazoxide-induced Ca2+ influx was not significantly inhibited by either voltage-operative Ca2+ channel blockers (nifedipine or verapamil), or by inhibitors of Na+, Ca2+-exchanger (bepridil and benzamil), but it was inhibited by flufenamic acid (FA), a Ca2+-permeable nonselective cation channel blocker. A quantitative analysis of apoptosis by flow cytometry revealed that a treatment with either FA or BAPTA, an intracellular Ca2+ chelator, significantly inhibited the diazoxide-induced apoptosis. Taken together, these results suggest that the observed diazoxideinduced apoptosis in the HepG2 cells may result from a Ca2+ influx through the activation of Ca2+-permeable non-selective cation channels. These results are very significant, and they lead us to further suggest that diazoxide may be valuable for the therapeutic intervention of human hepatomas.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams, J. M., and Cory, S., The Bcl-2 protein family: arbiters of cell survival.Science, 281, 1322–1326 (1998).
Aguilar-Bryan, L., Clement, J. P. 4th, Gonzalez, G., Kunjilwar, K., Babenko, A., and Bryan, J., Toward understanding the assembly and structure of KATP channels.Physiol. Rev., 78, 227–245 (1998).
Avdonin, V., Kasuya, J., Ciorba, M. A., Kaplan, B., Hoshi, T., and Iverson, L., Apoptotic proteins Reaper and Grim induce stable inactivation in voltage-gated K+ channels.Proc. Natl. Acad. Sci. U. S. A, 95, 11703–11708 (1998).
Barbiero, G., Duranti, F., Bonelli, G., Amenta, J. S., and Baccino, F. M., Intracellular ionic variations in the apoptotic death of L cells by inhibitors of cell cycle progression.Exp. Cell Res., 217, 410–418 (1995).
Bombeli, T., Karsan, A., Tait, J. F., and Harlan, J. M., Apoptotic vascular endothelial cells become procoagulant.Blood, 89, 2429–2442 (1997).
Bonnefoy-Berard, N., Genestier, L., Flacher, M., and Revillard, J. P., The phosphoprotein phosphatase calcineurin controls calcium-dependent apoptosis in B cell lines.Eur. J. Immunol., 24, 325–329 (1994).
Bortner, C. D., Hughes, F. M., Jr, and Cidlowski, J. A., A primary role for K+ and Na+ efflux in the activation of apoptosis.J. Biol. Chem., 272, 32436–32442 (1997).
Cameron, J. S., Lhuillier, L., Subramony, P., and Dryer, S. E., Developmental regulation of neuronal K+ channels by target-derived TGFin vivo andin vitro.Neuron, 21, 1045–1053 (1998).
Challinor-Rogers, J. L., and McPherson, G. A., Potassium channel openers and other regulators of KATP channels.Clin. Exp. Pharmacol. Physiol., 21, 583–597 (1994).
Chen, W. H., Yeh, T. H., Tsai, M. C., Chen, D. S., and Wang, T. H., Characterization of Ca2+-andvoltage-dependent nonselective cation channels inhumanHepG2 cells.J. Formos. Med. Assoc., 96, 503–510 (1997).
Chin, L. S., Park, C. C., Zitnay, K. M., Sinha, M., DiPatri, A. J. Jr., Perillan, P., and Simard, J.M., 4-Aminopyridine causes apoptosis and blocks an outward rectifier K+ channel in malignant astrocytoma cell lines.J. Neurosci. Res., 48, 122–127 (1997).
Cohen, J. J., and Duke, R. C., Glucocorticoid activation of a calcium-dependent endonuclease in thymocyte nuclei leads to cell death.J. Immunol., 132, 38–42 (1984).
Crompton, N. E., Programmed cellular response in radiation oncology.Acta Oncol., 37 Suppl 11, 1–4 (1998).
Deutsch, C., Potassium channels:basic functionand therapeutic aspects.Prog. Clin. Biol. Res., 334, 251–271 (1990).
Distelhorst, C.W., and Dubyak, G., Roleof calciumin glucocorticosteroid-induced apoptosisof thymocytesand lymphoma cells: resurrection of old theories by new findings.Blood, 91, 731–734 (1998).
Duchen, M. R., Roles of mitochondria in health and disease.Diabetes, 53, S96–102 (2004).
Edwards, G., and Weston, A.H., The role of potassium channels in excitable cells.Diabetes Res. Clin. Pract., 8 Suppl, S57–66 (1995).
Fasolato, C., Hoth, M., Matthews, G., and Penner, R., Ca2+ and Mn2+influxthrough receptor-mediatedactivationof nonspecific cation channels in mast cells.Proc. Natl. Acad. Sci. USA, 90, 3068–3072 (1993).
Fesus, L., Thomazy, V., and Falus, A., Induction and activation of tissue transglutaminase during programmed cell death.FEBS Lett., 224, 104–108 (1987).
Gogelein, H., Dahlem, D., Englert, H. C., and Lang, H. J., Flufenamic acid, mefenamic acid and niflumic acid inhibit singlenonselective cation channelsin therat exocrine pancreas.FEBS Lett., 268, 79–82 (1990).
Grynkiewicz, G., Poene, M., and Tsien, R. Y., A new generation of Ca2+ indicators withgreatly improved fluorescence properties.J. Biol. Chem., 260, 3440–3450 (1985).
Harman, A. W., and Maxwell, M. J., An evaluation of the role of calcium in cell injury.Annu. Rev. Pharmacol. Toxicol., 35, 129–144 (1995).
Hille, B., Ionic Channels of Excitable Membranes, 2nd Ed., Sinauer Associates, Inc., Sunderland, MA, pp. 115–139 and 472–503 (1992).
Hughes, F. M., Jr, Bortner, C. D., Purdy, G. D., and Cidlowski, J. A., Intracellular K+suppresses the activation of apoptosis in lymphocytes.J. Biol. Chem., 272, 30567–30576 (1997).
Jordan, J., Galindo, M. F., and Miller, R. J., Role of calpain and interleukin-1 beta converting enzyme-like proteases in the beta-amyloid-induced death of rat hippocampal neurons in culture.J. Neurochem., 68, 1612–1621 (1997).
Kamesaki, H., Mechanisms involved in chemotherapy-induced apoptosis and theirimplications in cancer chemotherapy.Int. J. Hematol., 68, 29–43 (1998).
Kamleiter, M., Hanemann, C. O., Kluwe, L., Rosenbaum, C., Wosch, S., Mautner, V. F., Muller, H. W., and Grafe, P., Voltage-dependent membrane currents of cultured human neurofibromatosis type 2 Schwann cells.Glia, 24, 313–322 (1998).
Kastan, M. B., Canman, C. E., and Leonard, C. J., P53, cell cycle control and apoptosis: implications for cancer.Cancer Metastasis Rev., 14, 3–15 (1995).
Kidd, V. J., Proteolytic activities that mediate apoptosis.Annu. Rev. Physiol., 60, 533–573 (1998).
Kim, J. A., Kang, Y. S., Lee, S. H., Lee, E. H., and Lee, Y. S., Involvement of Ca2+ influx in the mechanism of tamoxifen-induced apoptosis in HepG2 human hepatoblastoma cells.Cancer Lett., 147, 115–123 (1999).
Kim, J. A., Kang, Y. S., Lee, S. H., and Lee, Y. S., Inhibitors of Na+/Ca2+ exchanger prevent oxidant-induced intracellular Ca2+ increase andapoptosis in a human hepatoma cell line.Free Radic. Res., 33, 267–277 (2000).
Kornblau, S. M., The role of apoptosis in the pathogenesis, prognosis, and therapy of hematologic malignancies.Leukemia, 12 Suppl 1, S41–46 (1998).
Lepple-Wienhues, A., Berweck, S., Bohmig, M., Leo, C. P., Meyling, B., Garbe, C., and Wiederholt, M., K+ channels and the intracellular calcium signal in human melanoma cell proliferation.J. Membr. Biol., 151, 149–157 (1996).
Liu, S. I., Chi, C. W., Lui, W. Y., Mok, K. T., Wu, C. W., and Wu, S. N., Correlation of hepatocyte growth factor-induced proliferation and calcium-activated potassium current in human gastric cancer cells.Biochim. Biophys. Acta, 1368, 256–266 (1998).
Malhi, H., Irani, A. N., Rajvanshi, P., Suadicani, S. O., Spray, D. C., McDonald, T. V., andGupta, S., KATP channels regulate mitogenically-induced proliferationin primary hepatocytes and human liver cell lines: implications for liver growth control and potential therapeutic targeting.J. Biol. Chem., 275, 26050–26057 (2000).
McConkey, D. J. and Orrenius, S., The role of calcium in the regulation of apoptosis.J. Leukoc. Biol., 59, 775–783 (1996).
Melino, G., Annicchiarico-Petruzzeli, M., Piredda, L., Candi, E., Gentile, V., Davies, P. J., and Piacentini, M., Tissue transglutaminase and apoptosis: Sense and antisense transfection studies with human neuroblastoma cells.Mol. Cell Biol., 14, 6584–6596 (1994).
Minta, A. and Tsien, R. Y., Fluorescent indicators for cytosolic sodium.J. Biol. Chem., 264, 19449–19457 (1989).
Nilius, B. and Wohlrab, W., Potassium channels and regulation of proliferation of human melanoma cells.J. Physiol (Lord)., 445, 537–548 (1992).
Rouzaire-Dubois, B. and Dubois, J. M., K+ channel block-induced mammalian neuroblastoma cell swelling: a possible mechanism to influence proliferation.J. Physiol (Lond)., 510, 93–102 (1998).
Rouzaire-Dubois, B. and Dubois, J. M., A quantitative analysis of the role of K+ channels in mitogenesis of neuroblastoma cells.Cell Signal., 3, 333–339 (1991).
Schulte-Hermann, R., Bursch, W., Low-Baselli, A., Wagner, A., and Grasl-Kraupp, B., Apoptosis in the liver and its role in hepatocarcinogenesis.Cell Biol. Toxicol., 13, 339–348 (1997).
Shibasaki, F., Kondo, E., Akagi, E., and McKeon, F., Suppresion of signaling through NF-AT by interactions between calcineurin and BCL-2.Nature, 386, 728–731 (1997).
Shirihai, O., Attali, B., Dagan, D., and Merchav, S., Expression of two inward rectifier potassium channels is essential for differentiation of primitive human hematopoietic progenitor cells.J. Cell Physiol., 177, 197–205 (1998).
Squier, M. K. T. and Cohen, J. J., Calpain, an upstream regulator of thymocyte apoptosis.J. Immunol., 158, 3690–3697 (1997).
Squier, M. K. T., Miller, A. C. K., Malkinson, A. M., and Cohen, J. J., Calpain activationin apoptosis.J. Cell Physiol., 159, 229–237 (1994).
Stanley, R. G., Advances in second messenger and phosphoprotein research. Academic Press, Orlando, FL 33, 107–127 (1999).
Teshima, Y., Akao, M., Li, R. A., Chong, T. H., Baumgartner, W. A., Johnston, M. V., and Marban, E., Mitochondrial ATP- sensitive potassium channel activation protectacerebellar granule neurons from apoptosis induced by oxidative stress.Stroke, 34, 1796–1802 (2003).
Vermes, I., Haanen, C., Steffens-Nakken, H., and Reutelingsperger, C., A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V.J. Immunol. Methods, 184, 39–51 (1995).
Wang, S., Melkoumian, Z., Woodfork, K. A., Cather, C., Davidson, A. G., Wonderlin, W.F., and Strobl, J. S., Evidence for an early G1 ionic event necessary for cell cycle progression and survival in the MCF-7 human breast carcinoma cell line.J. Cell Physiol., 176, 456–464 (1998).
Wondergem, R., Cregan, M., Strickler, L., Miller, R., and Suttles, J., Membrane potassium channels and human bladder tumor cells: II. Growth properties.J. Membr. Biol., 161, 257–262 (1998).
Woodfork, K. A., Wonderlin, W. F., Peterson, V. A., and Strobl, J., Inhibition of ATP-sensitive potassium channels causes reversible cell-cycle arrest of human breast cancer cells in tissue culture.J. Cell Physiol., 162, 163–171 (1995).
Wyllie, A. H., Morris, R. G., Smith, A. L., and Dunlop, D., Chromatin cleavage inapoptosis: Association with condensed chromatin morphology and dependence ormacromolecular synthesis.J. Pathol., 142, 67–67 (1984).
Xu, B., Wilson, B. A., and Lu, L., Induction of human myeloblasts ML-1 cell G1 arrest by suppression of K+ channel activity.Am. J. Physiol., 271, C2037–2044 (1996).
Yu, S. P., Farhangrazi, Z. S., Ying, H. S., Yeh, C. H., and Choi D. W., Enhancement of outward potassium current may participate in β-amyloidpeptide-induced cortical neuronal death.Neurobiol. Dis., 5, 81–88 (1998).
Zhang, H., Inazu, M., Weir, B., Buchanan, M., and Daniel, E., Cyclopiazonic acid stimulates Ca2+ influxthrough non-specific cation channels in endothelial cells.Eur. J. Pharmacol., 251, 119–125 (1994).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, Y.S. Mechanism of apoptosis induced by diazoxide, a K+ channel opener, in HepG2 Human hepatoma cells. Arch Pharm Res 27, 305–313 (2004). https://doi.org/10.1007/BF02980065
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02980065