Abstract
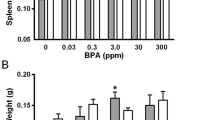
Bisphenol A [2, 2 bis (4-hydoxyphenyl) propane; BPA] is a widely used endocrine disruptors and has estrogenic activities. Although interests on biological effect of BPA are rising, evidences of its effect on immune system are lacking. We investigated that the effect of BPA on immune parameters to postulate the mechanism, and BPA interruptions between neuroendocrine and immune system. BPA was administrated to mice by p.o. (as a drinking water) dose on 0.015, 1.5 and 30 mg/ml for 4 weeks. The BPA treatment did not result in any change in body weight, spleen weight and distribution of lymphocyte subpopulation collected from spleen. BPA induced prolactin production in spleen, and exposure of BPA increased the activity of splenocyte proliferation in response to Con A (p<0.001). The production of a strong Th-1 type cytokine (IFN-γ) was induced while Th-2 type (IL-4) was suppressed by BPA treatment. These were consistent with RT-PCR results of transcription factor GATA-3 and IRF-1. These findings suggested that stimulation of prolactin production by estrogenic effects of BPA would affect cytokine profiles, and lead to imbalanced cellular immune response. In addition, we could speculate that prolactin and cytokine is important mediator involved in network between neuroendocrine and immune system by BPA.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Blake, C. A. and Boockfor, F. R., Chronic administration of the environmental pollutant 4-tert octylphenol to adult male rats interferes with the secretion of lutenizing hormone, follicle-stimulating hormone, prolactin and testosterone.Biology of Reproduction, 57, 255–266 (1997).
Bole-feysot, C., Goffin, V., Edery, M., Binart, N., Prolactin (PRL) and its receptor: action, signal transduction pathways and phenotypes observed in PRL receptor knockout Mice.Endocrine Reviews, 19 (3), 225–268 (1998).
Chun, T. Y. and Gorski, J., High concentrations of bisphenol A induce cell growth and prolactin secretion in an estrogen-responsive pituitary tumor cell line.Toxicology and Applied Pharmacology, 162, 161–165 (2000).
Clevenger, C. V., Russell, D. H., Appasamy, P. M. and Prystowsky, M. B., Regulation of interleukin 2-driven T-lymphocyte proliferation by prolactin.Proc. Natl. Acad. Sci., 87, 6460–6464 (1990).
Cook, J. C., Michael Kaplan, A., Davis, L. G. and O’Connor, J. C., Development of a tier I screening battery for detecting endocrine-active compound (EACs).Regulatory Toxicology and Pharmacology, 26, 60–68 (1997).
Dogusan, Z., Hooghe, R., Verdood, P., Hooghe-Peters, E. L., Cytokine-like effects of prolactin in human mononuclear and polymorphonuclear leukocytes.J. Neuroimmuno., 120, 58–66 (2001).
Feng, J. C., Loh, T. T. and Sheng, H. P., Lactation increases prolactin receptor expressions in spleen and thymus of rats.Life Sciences, 63 (2), 111–119 (1998).
Gala, R. R. and Rillema, J. A., Evaluation of prolactin-link activity produced by concanavalin-A stimulated mouse splenocytes.Life Sciences, 57 (18), 1683–1700 (1995).
Gala, R. R. and Shevach, E. M., Influence of bromocriptine administration to mothers on the development of pup thymocyte and splenocyte subsets and mitogen-induced proliferation in the mouse.Life Sciences, 53, 1981–1994 (1993).
Goloubkova, T., Ribeiro, M. F. M., Rodrigues, L. P., Cecconello, A. L., Spritzer, P. M., Effects of xenoestrogen bisphenol A on uterine and pituitary weight, serum prolactin levels and im-munoreactive prolactin cells in ovariectomized wistar rat.Arch. Toxicol., 74, 92–98 (2000).
Gunes, H. and Mastro, A. M., Prolactin receptor gene expression in rat splenocytes and thymocytes from birth to adulthood.Molecular and Cellular Endocrinology, 117, 41–52 (1996).
Imada, K., Leonard, W. J., The Jak-STAT pathway.Molecular Immunology, 37, 1–11 (2000).
Kavlock, R. J., Daston, G. P., Derosa, C., Fenner-Crisp, P., Gray, L. E., Kaattary, S., Lucier, G., Luster, M., Mac, M. J., Maczka, C., Miller, R., Moore, J., Rolland, R., Scott, G., Sheehan, D. M., Sink, T., Tilson, H. A., Research needs for the risk assessment of health and environmental effects of endocrine disruptors: A report of the U.S. EPA-sponsored workshop.Environmental Health Perspectives, 104, 1–26 (1996).
Ladics, G. S., Smith, C., Nicastro, S. C., Loveless, S. E., Cook, J. C. and O’Connor, J. C., Evaluation of primary humoral immune response following exposure of male rats to 17β-estradiol or flutamide for 15 days.Toxicological Sciences, 46, 75–82 (1998).
Lomnee, H., Jouventin, P., Lacroix, A., Lallemand, J. and Chastel, O., Reproductive endocrinology of tropical seabirds: Sex-specific patterns in LH, steroids, and prolactin secretion in relation to parental care. General and comparative.Endocrinology, 117, 413–426 (2000).
Masten, S. A. and Shiverick, K. T., Evidence that a mitogen-inducible prolactin-immunoreactive protein in rat spleen lymphocytes is aldolase A.Life Sciences, 60 (24), 2173–2182 (1997).
Matera, L., Cutufia, M., Geuna, M., Contarini, M., Buttiglieri, S., Galin, S., Fazzari, A., Cavaliere, C., Prolactin is an autocrine growth factor for the Jurkat human T-leukemic cell line.Journal of Neuroimmunology 79, 12–21 (1997).
Matera, L., Mori, M., Geuna, M., Buttiglieri, S., Palestra, G., Prolactin in autoimmunity and antitumor defense.Journal of Neuroimmunology, 109, 47–55 (2000).
McMurrary, R. W., Estrogen, prolactin, and autoimmunity: actions and interactions.International Immunophamacology, 1, 995–1008 (2001).
Mukherjee, P., Mastro, A. M. and Hymer, W. C., Prolactin induction of interleukin-2 receptor on rat splenic lymphocytes.Endocrinology, 126 (1), 88–94 (1990).
Nikula, H., Talonpoika, T., Kaleva, M. and Toppari, J., Inhibition of hCG-stimulated steroidogenesis in cultured mouse leydig tumor cells by bisphenol A and octylphenols.Toxicology and Applied Pharmacology, 157, 166–173 (1999).
Postel-Vinay, M. C., Coelho, V. de M., Gagnerault, M. C. and Dardenne, M., Growth hormone stimulates the proliferation of activated mouse T lymphocytes.Endocrinology, 138 (5), 1816–1820 (1997).
Romagnani, S., The Th1/Th2 paradigm.Immunology Today 18 (6), 263–266 (1997).
Steinmetz, R., Brown, N.G., Allen, D.L., Bigsby, R.M. and Ben-Jonathan, N., The environmental estrogen bisphenol A stimulates prolactin releasein vitro andin vivo.Endocrinology 138 (5), 1780–1786 (1997).
Stoker, T. E., Robinette, C. L., Britt, B. H., Laws, S. C. and Cooper, R. L., Prepubertal exposure to compounds that increase prolactin secretion in the male rat: effect on the adult prostate.Biology of Reproduction, 61, 1636–1643 (1999).
Takahashi, S., Chi, X. J., Yamaguchi, Y., Suzuki, H., Sugaya, S., Kita, K., Hiroshima, K., Yamamori, H., Ichinose, M. and Suzuki, N., Mutagenicity of bisphenol A and its suppression by interferon-/’a in RSa cells.Mutation Research, 490, 199–207 (2001).
Takao, T., Nanamiya, W., Nagano, I., Asaba, K., Kawabata, K. and Hashimoto, K., Exposure with the environmental estrogen bisphenol A disrupts the male reproductive tract in young mice.Life Sciences, 65 (22), 2351–2357 (1999).
Takeuchi, T. and Tsutsumi, O., Serum bisphenol A concentrations showed gender differences, possibly, linked to androgen levels.Biochemical and Biophysical Research Communications, 291, 76–78 (2002).
Yamagata, T., Mitani, K., Oda, H., Suzuki, T., Honda, H., Asai, T., Maki, K., Nakamoto, T., Hiral, H., Acetylation of GATA-3 affects T-cell survival and homing to secondary lymphoid organs.The EMBO Journal, 19 (17), 4676–4687 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Youn, J.Y., Park, H.Y., Lee, J.W. et al. Evaluation of the immune response following exposure of mice to bisphenol A: Induction of th1 cytokine and prolactin by BPA exposure in the mouse spleen cells. Arch Pharm Res 25, 946–953 (2002). https://doi.org/10.1007/BF02977018
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02977018