Abstract
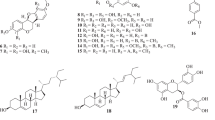
In this study, we isolated two new isorhamnetin glycosides, designated as nelumboroside A (3) and nelumboroside B (4), as well as the previously-characterized isorhamnetin glucoside (1) and isorhamnetin rutinoside (2), from then-BuOH fraction ofNelumbo nucifera stamens. The structures of the two new compounds were then determined, using chemical and spectroscopic techniques. All isolated isorhamnetin glycosides1–4 showed marked antioxidant activities in the DPPH, and ONOO− assays.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Blois, M. S., Antioxidant determination by the use of a stable free radical.Nature, 181, 1199–1202 (1958).
Furukawa, H., On the alkaloids ofNelumbo nucifera Gaertn. XII. Alkaloids of loti embryo. (5).Yakugaku Zasshi, 86, 75–77 (1966a).
Furukawa, H., Studies on the alkaloids ofNelumbo nucifera Gaertn. NMR spectra of liensinine type alkaloids.Yakugaku Zasshi, 86, 883–886 (1966b).
Furukawa, H., Yang, T. H., and Lin, T. J., On the alkaloids ofNelumbo nucifera Gaertn. XI. Alkaloids of loti embryo. (4). Structure of lotusine, a new water-soluble quaternary base.Yakugaku Zasshi, 85, 472–475 (1965).
Hörhammer, L., Wagner, H., Amdt, H. G., and KraemerL.Farkas, H., Synthese natürlich vorkommender polyhydroxy-flavonol-glykoside.Tetrahedron Lett., 7, 567–571 (1966).
Jung, H. A., Kim, J. E., Chung, H. Y., and Choi, J. S., Antioxidant principles ofNelumbo nucifera stamens.Arch. Pharm. Res., 26, 279–285 (2003).
Kim, J. G., Illustrated Natural Drugs Encyclopedia (color edition). Namsandong, Korea, Vol. II, p. 95 (1997).
Kim, T. J., Korean Resources Plants. Seoul National University Publisher, Seoul, Korea, Vol. II, pp. 218–219 (1996).
Kooy, N. W., Royall, J. A., Ischiropoulos, H., and Beckman, J. S., Peroxynitrite-mediated oxidation of dihydrorhodamin 123.Free Radical Biol. Med., 16, 149–156 (1994).
Koshiyama, H., Ohkuma, H., Kawaguchi, H., Hsü H. Y., and Chen, Y. P., Isolation of 1-(p-hydroxybenzyl)-6,7-dihydroxy-1,2,3,4-tetrahydroisoquinoline (demethylcoclaurine), an active alkaloid fromNelumbo nucifera.Chem. Pharm. Bull., 18, 2564–2568 (1970).
Kunitomo, J. I., Miyoshi, M., Maruyama, T., and Yuge, E., Studies on the alkaloids ofNelumbo nucifera Gaertn. XIV. Synthesis ofdl-nornuciferine (dl-1-methoxyaporphine).Yakugaku Zasshi, 91, 896–900 (1971).
Kunitomo, J. I., Nagai, Y., Okamoto, Y., and Furukawa, H., Studies on the alkaloids ofNelumbo nucifera Gaertn. XIV. On the tertiary base.Yakugaku Zasshi, 90, 1165–1169 (1970).
La Cour, B., Molgaard, P., and Yi, Z., Traditional Chinese medicine in treatment of hyperlipidaemia.J. Ethnopharmacol., 46, 125–129 (1995).
Lee, M. W., Kim, J. S., Cho, S. M., Kim, J. H., and Lee, J. S., Anti-diabetic constituent from the node of lotus rhizome (Nelumbo nucifera Gaertn).Natural Product Science, 7, 107–109 (2001).
Ling, Z. Q., Xie, B. J., and Yang, E. L., Isolation, characterization, and determination of antioxidative activity of oilgomeric procyanidins from the seedpod ofNelumbo nucifera Gaertn.J. Agric. Food Chem., 53, 2441–2445 (2005).
Mabry, T. J., Markham, K. R., and Thomas, M. B., The Systematic Identification of Flavonoids. Springer-Verlag, Berlin, Chapter 1 (1970).
Markham, K. R., Techniques of Flavonoid Identification. Academic Press, London, U. K. (1982).
Markham, K. R., Ternai, B., Stanley, R., Geiger, H., and Mabry, T. J., Carbon-13 NMR studies of flavonoids-III: Naturally occurring flavonoid glycosides and their acylated derivatives.Tetrahedron, 34, 1389–1397 (1978).
Mukherjee, P. K., Pal, S. K., Saha, K., and Saha, B. P., Hypoglycaemic activity ofNelumbo nucifera Gaertn (Fam. Nymphaeaceae) rhizome (Methanolic extract) in streptozotocin-induced diabetic rats.Phytother. Res., 9, 522–524 (1995).
Mukherjee, P. K., Saha, K., Pal, M., and Saha, B. P., Effect ofNelumbo nucifera rhizome extract on blood sugar level in rats.J. Ethnopharmacol., 58, 207–213 (1997).
Sung, S. H., Jung, W. J., and Kim, Y. C., A novel flavonol lyxoside ofOrostashys japonicus herb.Natural Product Letters, 16, 29–32 (2002).
Tomita, M., Furukawa, H., Yang, T. H., and Lin, T. J., On the alkaloids ofNelumbo nucifera Gaertn. VIII. Studies on the alkaloids of loti embryo. (1). Structure of isoliensinine, a new biscoclaurine type alkaloid.Chem. Pharm. Bull., 13, 39–43 (1965).
Van Bergen, P. F., Hatcher, P. G., Boon, J. J., Collinson, M. E., and De Leeuw, J. W., Macromolecular composition of the propagule wall ofNelumbo nucifera.Phytochemistry, 45, 601–617 (1997).
Wu, S., Sun, C., Cao, X., Zhou, H., Zhang, H., and Pan, Y., Preparative counter-current chromatography isolation of liensinine and its analogues from embryo of the seed ofNelumbo nucifera Gaertn. Using upright coil planet centrifuge with four multilayer coils connected in series.J. Chromatogr. A., 1041, 153–162 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hyun, S.K., Jung, Y.J., Chung, H.Y. et al. Isorhamnetin glycosides with free radical and ONOO− scavenging activities from the stamens ofNelumbo nucifera . Arch Pharm Res 29, 287–292 (2006). https://doi.org/10.1007/BF02968572
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02968572