Abstract
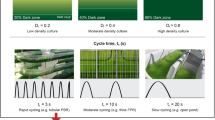
The physiological characteristics of cultures of very high cell mass (e.g. 10 g cell mass/L), termed “ultrahigh cell density cultures” is reviewed. A close relationship was found between the length of the optical path (OP) in flat-plate reactors and the optimal cell density of the culture as well as its areal (g m−2 day−1) productivity. Cell-growth inhibition (GI) unfolds, as culture density surpasses a certain threshold. If it is constantly relieved, a 1.0 cm OP reactor could produceca. 50% more than reactors with longer OP,e.g. 5 or 10 cm. This unique effect, discovered by Hu et al. [3], is explained in terms of the relationships between the frequency of the light-dark cycle (L-D cycle), cells undergo in their travel between the light and dark volumes in the reactor, and the turnover time of the photosynthetic center (PC). In long OP reactors (5 cm and above) the L-D cycle time may be orders of magnitude longer than the PC turnover time, resulting in a light regime in which the cells are exposed along the L-D cycle, to long, wasteful dark periods. In contrast, in reactors with an OP ofca. 1.0 cm, the L-D cycle frequency approaches the PC turnover time resulting in a significant reduction of the wasteful dark exposure time, thereby inducing a surge in photosynthetic efficiency. Presently, the major difficulty in mass cultivation of ultrahigh-density culture (UHDC) concerns cell grwoth inhibition in the culture, the exact nature of which is awaiting detailed investigation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Javanmardian, M. and B. O. Palsson (1991) High-density photoautrotrophic algal cultures: Design, construction, and operation of a novel photobioreactor system.Biotechnol. Bioeng. 38: 1182–1189.
Hu, Q., Y. Zarmi, and A. Richmond (1998) Combined effects of light intensity, light-path, and culture density on output rate ofSpirulina platensis (Cyanobacteria).Eur. J. Phycol. 33: 165–171
Hu, Q., H. Guterman, and A. Richmond (1996) A flat modular photobioreactor (FIMP) for outdoor mass cultivation of photoautotrophs.Biotechnol. Bioeng. 51: 51–60.
Richmond, A., C-W. Zhang, and Y. Zarmi (2003) Efficient use of strong light for high photosynthetic productivity: Interrelationships between the optical path, the optimal population density and cell-growth inhibition. In:Biomolecular Engineering, Special issue of the Conference of the European Society for Marine Biotechnology “Marine Biotechnology: Basics and applications”. 20: 229–236.
Janssen, M., J. Tramper, L. R. Mur, and R. H. Wijffels (2003) Enclosed outdoor photobioreactors: Light regime, photosynthetic efficiency, scale-up, and future prospects.Biotechnol. Bioeng. 81: 193–210.
Dubinsky, Z., P. G. Falkowsky, and K. Wyman (1986) Light harvesting and utilization by phytoplankton.Plant Cell Physiol. 27: 1335–1349.
Dubinsky, Z. (1992) The functional and optical absorption cross-sections of phytoplankton photosynthesis. pp. 31. In:Primary Productivity and Biogeochemical Cycles in the Sea. Plenum Press, New York, USA.
Zarmi, Y: (2003) Personal communication.
Hu, O., H. Guterman, and A. Richmond (1996b) Physiological characteristics ofSpirulina platensis cultured in ultra-high cell densities.J. Phycol. 32: 1066–1073
Sukenik, A., R. S. Levy, Y. Levy, P. G. Falkowsky, and Z. Dubinsky (1991) Optimizing algal biomass production in an outdoor pond: A simulation model.J. Appl. Phycol. 3: 191–201.
Richmond, A. (2003)Handbook of Microalgae Culture: biotechnology and Applied Phycology. p. 575. Blackwell Books, Oxford, UK.
Vonshak, A. and R. Guy (1992) Photoadaptation, photoinhibition and productivity in the blue-green alga.Spirulina platensis grown outdoors.Plant Cell Environ. 15: 613–616.
Torzillo, G., A. J. Komend, J. Kopecky, C. Faraloni, and J. Masojidek (2003) Photoinhibitory stress induced by high oxygen and low temperature in outdoor cultures ofArthrospira platensis grown in closed photobioreactors. p. 31.Abstracts of Third European Phycological Congress. July 21–26. Belfast, Ireland.
Märkl, H. (1980) Modeling of algal production systems. In: G. Shelef, and C. J. Soeder (eds.),Algal Biomass. Elsevier/North-Holland, Amsterdam, The Netherlands.
Richmond, A. and J. U. Grobbelaar (1986) Factors affecting the output rate ofSpirulina platensis with reference to mass cultivation.Biomass 10: 253–264.
Barbosa, M. J. (2003)Microalgal Photobioreactors: Scaleup and Optimization. Ph.D. Thesis. Wageningen University, Wageningen, The Netherlands.
Barbosa, M. J., M. Albrecht, and R. H. Wijffels (2003) Hydrodynamic stress and lethal events in sparged microalgae cultures.Biotechnol. Bioeng. 83: 112–120.
Miron, A. S., A. C. Gomez, F. G. Camacho, E. M. Grima, and M. Y. Chisti (1999) Comparative evaluation of compact photobioreactors for large-scale mono-culture of microalgae.J. Biotechnol. 70: 249–256.
Camacho, F. G., A. C. Gomez, T. M. Sobcruk, and E. M. Grima (2000) Effects of mechanical and hydrodynamic stress in agitated, sparged cultures ofPorphyridium cruentum.Proc. Biochem. 35: 1045–1050.
Pratt, R. (1942) Studies onChlorella vulgaris: V. Some of the properties of the growth inhibitors formed byChlorella cells.Amer. J. Bot. 29: 142–148.
Leving, T. (1945) Some culture experiments with marine plankton diatoms.Med. Oceanogr. Inst. Gotenborg 3: 12.
Von Dennffer, D. (1948) Übereinen Wachstum-Hemmstoff in älternden Diatomeenkulturen.Biol. Zentralbl. 67: 7–13.
Lefevre, M. (1964) Extracellular products of algae. pp 337–367. In: D. F. Jackson (ed.),Algae and Man. Plenum Press, New York, USA.
Fogg, G. E. (1971) Extracellular products of algae in fresh water.Arch. Hydrobiol. 5: 1–25.
Harris, D. O. (1975) Antibiotics production by the green alga,Pandorina morum. pp. 106–111. In: L. Brezonik and J. L. Fox (eds.),Water Quality Management through Biological Control. University of Florida, Gainesville, USA.
Keeting, K. I. (1978) Blue-green algal inhibition of diatom growth: Transition from mesotrophic to eutrophic community structure.Science 199: 971–973.
Pratt, R. and J. Fong (1940) Influence of the size of inoculum on the growth ofChlorella vulgaris in freshly prepared culture medium.Amer. J. Bot. 27: 52–56.
Curl, H. and G. C. McLeod (1961) The physiological ecology of a marine diatomSkeletonema costatum. (Grev.) Cleve. J. Mar. Res. 19: 70–88.
McCracken, M. D., R. E. Middaugh, and R. S. Middaugh (1980) A chemical characterization on an algal inhibitor obtained fromChlamydomonas.Hydrobiol. 70: 271–276
Imada, N., K. Kobayashi, K. Tahara, and Y. Oshima (1991) Production of an autoinhibitor bySkeletonema costatum and its effect on the growth of other phytoplankton.Nippon Suisan Gakkaishi 57: 2285–2290.
Imada, N., K. Kobayashi, K. Isomura, H. Saito, S. Kimura, K. Tahara, and Y. Oshima (1992) Isolation and identification of an autoinhibitor produced bySkeletonema costatum.Nippon Suisan Gakkaishi 58: 1687–1692.
Mandalam, R. K. and B. O. Palsson (1995)Chlorella vulgaris (Chlorellaceae) does not secrete autoinhibitors at high cell densities.Amer. J. Bot. 82: 995–963.
Mandalam, R. K. and B. O. Palsson (1998) Elemental balancing of biomass and medium composition enhances grwoth capacity in high-densityChlorella vulgaris cultures.Biotechnol. Bioeng. 59: 605–611.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Richmond, A. Growth characteristics of ultrahigh-density microalgal cultures. Biotechnol. Bioprocess Eng. 8, 349–353 (2003). https://doi.org/10.1007/BF02949278
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02949278