Abstract
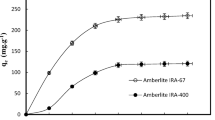
Weak-, moderate-, and strong-base resins were evaluated for their sorption capacities of lactic acid from solutions with different pHs. Composite isotherms and breakthrough curves indicated that the sorption capacities of weak- and moderate-base resins decreased markedly as the pH of the feed exceeded the pKa of lactic acid. The decrease in capacity was mainly the result of the decrease in concentration of undissociated lactic acid, the species preferentially adsorbed by the resin, as the pH of the feed increases. The sorption capacity of the strong-base resin remained constant in the same pH range (pH 2–6).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Lipinsky, E. S. and Sinclair, R. G. (1986),Chem. Eng. Prog. 82(8), 26–32.
Kabawata, N., Yoshida, J., and Tanigawa, Y. (1981),Ind. Eng. Chem. Prod. Res. Dev. 20(2), 386–390.
Chanda, M., O’Driscoll, K. F., and Rempel, G. L. (1985),Reactive Polymers 4, 39–48.
Kuo, Y., Munson, C. L., Rixey, W. G. A., Garcia, A. A., Frierman, M., and King, C. J. (1987),Sep. Purif. Methods 16(1), 31–64.
Munson, C. L., Garcia, A. A., Kuo, Y., Frierman, M., and King, C. J. (1987),Sep. Purif. Methods 16(1), 65–89.
Frierman, M., Kuo, Y., Joshi, D., Garcia, A. A., and King, C. J. (1987),Sep. Purif. Methods 16(1), 91–102.
Kalprathipanja, S. (1988), US Patent 4,720,579.
Kalprathipanja, S., Oroskar, A. R., and Priegnitz, G. W. (1989), US Patent 4,851,573.
Kalprathipanja, S. (1989), US Patent 4,851,574.
Garcia, A. A. and King, C. J. (1989),Ind. Eng. Chem. Res. 28(2), 204–212.
Davison, B. H. and Scott, C. D. (1991),Biotechnol. Bioeng. 39(3), 365–368.
Kalprathipanja, S. and Oroskar, A. R. (1991), US Patent 5,068,418.
King, C. J. and Tung L. A. (1992), US Patent 5,132,456.
Srivastava, A., Roychoudhury, P. K., and Sahai, V. (1992),Biotechnol. Bioeng. 39(6), 607–613.
Rossiter, G. J. and Tolbert, C. A. (1991), Paper presented at the AIChE National Meeting, Los Angeles, CA.
Ernst, E. E. and McQuigg, D. W. (1992), Paper presented at the AIChE National Meeting, Miami Beach, FL.
Weast, R. C., ed. (1987),Handbook of Chemistry and Physics, CRC Press, Boca Raton, FL, p. D-99.
Helfferich, F. (1962),Ion Exchange, McGraw-Hill, New York, pp. 231–232.
American Society of Testing and Materials Method Number E 203–75 (1975),Am. Soc. Test. Mater., Philadelphia, PA.
Rohm and Haas (1988),Rohm and Haas Ion Exchange Resin Laboratory Guide, Philadelphia, PA.
Kipling, J. J. (1965),Adsorption from Solution of Non-electrolytes, Academic Press, New York.
Ruthven, D. M. (1984),Principles of Adsorption and Adsorption Processes, Wiley and Sons, New York, pp. 86–91.
Maciel, G. E., Traficante, D. D., and Lavalle, D. (1978),Chemistry, D. C. Heath, Lexington, MA, p. 216.
Anderson, R. E. (1979), inHandbook of Separation Techniques for Chemical Engineering, P. A. Schweitzer, ed., McGraw-Hill, New York, pp. 1-379–1-381.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Evangelista, R.L., Mangold, A.J. & Nikolov, Z.L. Recovery of lactic acid by sorption. Appl Biochem Biotechnol 45, 131–144 (1994). https://doi.org/10.1007/BF02941793
Issue Date:
DOI: https://doi.org/10.1007/BF02941793