Abstract
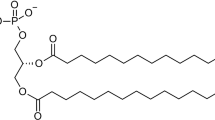
The phase transition behavior of 1, 2-di-n-heptadecanoyl phosphatidylcholine (DHPC) with and without water has been studied by use of differential scanning calorimetry. It was found by experiment that the glass transition occurred at first during the first heating of a sample of DHPC without water and then the sample underwent melting as an ordinary crystal. Therefore the sample of DHPC without water was a glassy crystal. However, the DHPC sample crystallizing from melt was an ordinary crystal. From the relationship between the total melting enthalpy Q1 of freezable water and the water contenth, it was concluded that the water contained in the DHPC samples might exist in three states recognizable thermodynamically. The water in the first state was an unfreezable water. It was the water bound directly with the head groups of the phospholipid, i.e. the primary hydration water. Every head group might bind seven such molecules of water. The water in the second state was the secondary hydration water, its melting point was close to that of pure water and its specific melting enthalpy dQr/dh was lower than that of pure water. Every molecule of DHPC might possess at most about six such molecules of water. With the further increasing of the water content, the bulk water appeared in the system, its melting point and specific melting enthalpy were close to those of pure water. The experiments demonstrated that the melting peak of DHPC shifted toward the side of lower temperature with the increasing of water content. Ath>16.2 (percent by weight), the temperature Ttr and the enthalpy Δtr of the gel-liquid crystal phase transition of the lipid were unchanged substantially with the increasing of water content. The experimental values of the quantities,T tr and Δtr, and phase transition entropy Δtr were in good agreement with those calculated by the set of Huang equations (1981).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ladbrooke, B. D., Chapman, D., Thermal analysis of lipids, proteins and biological membranes: Review and summary of some recent studies,Chem. Phys. Lipids, 1969, 3(4): 304.
Kodama, M., Kuwabara, M., Seki, S., Successive phase-transition phenomena and phase diagram of the phosphatidylcholine-water system as revealed by differential scanning calorimetry,Biochim. Biophys. Acta, 1982, 689(3): 567.
Lewis, R. N. A. H., Mak, N., McElhaney, R. N., A differential scanning calorimetric study of the thermotropic phase behavior of model membranes composed of phosphatidylcholines containing linear saturated fatty acyl chains,Biochemistry, 1987, 26(19): 6118.
Privalov, P. L., Stability of proteins: Proteins which do not present a single cooperative system, inAdvances in Protein Chemistry (ed. Anfinsen, C. B.), New York: Academic Press, 1982, 1–104.
Sturtevant, J. M., Biochemical applications of differential scanning calorimetry,Annu. Rev. Phys. Chem., 1987, 38: 463.
Wang Bangning, Tan Fu, A study on the interaction of proteinase K and water by low temperature calorimetry,Chinese J. Biochem. Biophys. (in Chinese), 1992, 25(3): 213.
Chen, S. C., Sturtevant, J. M., Gaffney, B. J., Scanning calorimetric evidence for a third phase transition in phosphatidylcholine,Proc. Natl. Acad. Sci. USA, 1980, 77(9): 5060.
Mason, J. T., Huang, C.-Hq, Chain length dependent thermodynamics of saturated symmetric-chain phosphatidylcholine bi-layers,Lipids, 1981, 16(8): 604.
Lewis, R. N. A. H., McElhaney, R. N., Calorimetric and spectroscopic studies of polymorphic phase behavior of a homologous series of n-saturated 1, 2-diacyl phophatidylethanolamines,Biophys. J., 1993, 64(4): 1081.
Lewis, R. N. A. H., McElhaney, R. N., Thermotropic phase behavior of model membranes composed of phosphatidylcholines containing isobranched fatty acids: 1. Differential scanning calorimetric studies,Biochemistry, 1985, 24(10): 2431.
Silvius, J. R., McElhaney, R. N., Effects of phospholipids acyl chain structure on physical properties: (I) Isobranched phosphatidylcholines,Chem. Phys. Lipids, 1979, 24(3): 287.
Silvius, J. R., McElhaney, R. N., Effects of phospholipid acyl chain structure on thermotropic phase transitions: 3. Phosphatidylcholines with (-) - and (f) -antiisoacyl chains,Chem. Phys. Lipids, 1980, 26(1): 67.
Small, D. M., Appendix VI phospholipids, inThe Physical Chemistry of Lipids: From Alkanes to Phospholipids, New York: Plenum Press, 1986, 627–635.
Author information
Authors and Affiliations
Additional information
Project supported by the National Natural Science Foundation of China.
Rights and permissions
About this article
Cite this article
Wang, B., Tan, F. DSC study of phase transition of anhydrous phospholipid DHPC and influence of water content. Sc. China Ser. B-Chem. 40, 412–418 (1997). https://doi.org/10.1007/BF02877758
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02877758