Abstract
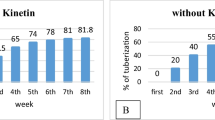
In vitro tuberization as a potential screening method for heat stress tolerance in potato, was assessed on nodal expiants of Desirée, LT-2, Kennebec and Russet Burbank. Two tuber inducing media protocols were evaluated at 20 C and 28 or 30 C. Independently of the media protocol, heat stress significantly reduced tuberization. A delay in the formation of tuber initials was also observed in Desirée, Kennebec and LT-2 at 28 and 30 C compared to 20 C. Russet Burbank failed to tuberize under heat stress on both media. At higher temperatures Desirée either did not tuberize, or tuberized poorly on high sucrose-agar medium and tuberized the best of all cultivars, on low sucrose-Gelrite medium. Kennebec and LT-2 tuberized on both media. Medium with Gelrite gave better tuberization and more reproducible results than with agar. A high sucrose-agar medium, on the other hand, separated the heat tolerant clone LT-2 from the other cultivars.
Higher temperature reduced accumulation of patatin and 22 kDa protein in all cultivars. The reduction was greater in Kennebec and least in LT-2. The results indicate that microtuber production under heat stress conditions, combined with SDS-PAGE protein electrophoresis, can be considered as a preliminary method in screening potato germplasm for subtropical and tropical climates.
Compendio
Se ha ensayado la tuberizaciónin vitro como un método potencial de tamizado para tolerancia al estrés por calor en papa, sobre explantas nodales de Desirée, LT-2, Kennebec y Russet Burbank. Se evaluaron dos medios de inducción de tuberización a 20 C y 28 ó 30 C. Independientemente del medio, el estrés por calor redujo significativamente la tuberización. Se observó también un retardo en la formación de iniciales del tubérculo en Desirée, Kennebec y LT-2 a 28 y 30 C comparado con 20 C. Russet Burbank no llegó a tuberizar bajo estrés por calor en ambos medios. Desirée no tuberizó o tuberizó muy poco en medio de agar con alto contenido de sucrosa y tuberizó mejor que todos los cultivares en medio Gerlita con bajo contenido de sucrosa. Kennebec y LT-2 tuberizaron en ambos medios. Ed medio con Gerlita dió mejor tuberización y resultados más reproducibles que el con agar. Por otra parte, un medio de agar con alto contenido de sucrosa diferenció al clon tolerante LT-2 de los otros cultivares.
Las temperaturas más altas reducen la acumulación de patatina y proteina 22 kDa en todos los cultivares. La reducción fué mayor en Kennebec y mucho menor en LT-2. Los resultados indican que la producción de microtubérculos bajo condiciones de estrés al calor, combinada con electroforesis de proteina SDS-PAGE, pueden ser considerados como un método preliminar para el tamizado de germoplasma de papa en climas tropicales y subtropicales.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Literature Cited
Borah, M.N. and F.L. Milthorpe. 1962. Growth of potato as influenced by temperature. Indian J P1 Physiol 5:53–72.
Bourque, J.E., J.C. Miller and W.D. Park. 1987. Use of anin vitro tuberization system to study tuber protein gene expression.In Vitro Cell Dev Biol 23:381–386.
Chua, N.H. 1980. Methods in Enzymology 69. Part C. Academic Press Inc., New York.
Ewing, E.E. 1981. Heat stress and the tuberization stimulus. Am Potato J 58:31–49.
Ewing, E.E. 1985. Cuttings as simplified models of the potato plant.In: P.H. Li (ed.). Potato Physiology, Academic Press Inc., Orlando, Fla. pp. 153–207.
Gautney, T.L. and F.L. Haynes. 1983. Recurrent selection for heat tolerance in diploid potatoes (Solarium tubervsum subsp.phureja andstenotomum). Am Potato J 60:537–542.
Hannapel, D.J., J.C. Miller and W.D. Park. 1985. Regulation of potato tuber protein accumulation by gibberellic acid. Plant Physiol 78:700–703.
Kheder, M.B. and E.E. Ewing. 1985. Growth and analysis of eleven potato cultivars grown in the greenhouse under long photoperiods with and without heat stress. Am Potato J 62:537–554.
Levy, D. 1984. CultivatedSolanum tuberosum L. as a source for the selection of cultivars adapted to hot climates. Tropical Agric (Trinidad) 61:167–170.
Liede, B.E., T. Rosier and S.L. Desborough. 1988. HPLC isolation and nutritive value of a major tuber protein. Am Potato J 64:545–557.
Machado, V.S. and J. Shupe. 1985.In vitro microtuberization of seed potatoes.In: Tissue Culture as a Plant Production System for Horticultural Crops, Beltsville, Maryland. Abs 17.
Marinus, J. and K.B.A. Bodlaender. 1975. Response of some potato varieties to temperature. Potato Res 18:189–204.
Midmore, D.J. 1984. Potato (Solarium spp.) in the hot tropics. I. Soil temperature effects on emergence, plant development and yield. Field Crops Res 8:255–271.
Paiva, E., R.M. Lister and W.D. Park. 1983. Induction and accumulation of major tuber proteins in stems and petioles. Plant Physiol 71:156–160.
Park, W.D., C. Blackwood, G.A. Mignery, M.A. Hermodson and R.M. Lister. 1983. Analysis of the heterogeneity of the molecular weight 40,000 tuber glycoprotein of potatoes by immunological methods and NH2-terminal sequence analysis. Plant Physiol 71:156–160.
Park, W.D. 1984. Potato tuber proteins as molecular probes for tuberization. Hort Science 19:37–40.
Racusen, D. and M. Foote. 1980. A major soluble glycoprotein of potato tubers. J Food Biochem 4:43–51.
Sattelmacher, B. 1983. A rapid seedling test for adaptation to high temperatures. Potato Res 26:133–138.
Sipos, J., J. Nowak and G. Hicks. 1988. Effect of daminozide on survival, growth and yield of micropropagated potatoes. Am Potato J 65:353–364.
Villamayor, F.G.J. 1983. Preliminary screening of potatoes for heat tolerance. Radix 5(2):6–7.
Wallevik, K. and J.C. Jensenius. 1982. A simple and reliable method for drying of polyacrylamide slab gels. J Biochem Biophys Methods 6:17–21.
Author information
Authors and Affiliations
Additional information
Project supported by the NSERC student award and N.S.D.A.M. operating grant No. 87-89.
Rights and permissions
About this article
Cite this article
Nowak, J., Colborne, D. In vitro tuberization and tuber proteins as indicators of heat stress tolerance in potato. American Potato Journal 66, 35–45 (1989). https://doi.org/10.1007/BF02853487
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02853487