Abstract
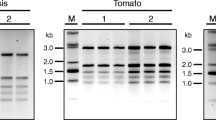
A protocol is described for rapid RNA isolation from various plant species and tissues rich in polyphenolics and polysaccharides. The method is based on the Nucleon PhytoPure™ system without the use of phenol. The procedure can be completed within 1 h and many samples can be processed at the same time. The yield ranged from 240 μg up to 3 mg per gram of tissue with an average purity measured as A260/280 of 1.85. The RNA was of sufficient quality for use in RT-PCR reactions. Quantitation of single-stranded cDNA was carried out with the RiboGreen™ reagent and of PCR products with the PicoGreen™ reagent.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CTAB:
-
cetyltrimethylammonium bromide
References
Ahn SJ, Costa J and Emanuel JR (1996) PicoGreen quantitation of DNA: effective evaluation of samples pre- or post-PCR. Nucleic Acids Res 24: 2623–2625.
Ainsworth C (1994) Isolation of RNA from floral tissue ofRumex acetosa (Sorrel). Plant Mol Biol Reptr 12: 198–203.
Chang S, Puryear J and Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Reptr 11: 113–116.
Claros MG and Cánovas FM (1998) Rapid high quality RNA preparation from pine seedlings. Plant Mol Biol Reptr 16: 9–18.
De Vries S, Hoge H and Bisseling T (1993) Isolation of total and polysomal RNA from plant tissues. In: Gelvin SB, Schilperoort RA and Verma DPS (eds.), Plant Molecular Biology Manual, Kluwer Academic Publishers, Dordrecht, B6, pp. 1–13.
Dong J-Z and Dunstan DI (1996) A reliable method for extraction of RNA from various conifer tissues. Plant Cell Rep 15: 516–521.
Ernst D, Kiefer E, Heller W and Sandermann H (1999) RNA-Isolierungs-Kit. Patent applied for Deutsches Patentamt (199 16 534.3).
Geuna F, Hartings H and Scienza A (1998) A new method for rapid extraction of high quality RNA from recalcitrant tissues of grapevine. Plant Mol Biol Reptr 16: 61–67.
Grosskopf E, Wegener-Strake A, Sandermann H and Ernst D (1994) Ozone-induced metabolic changes in Scots pine: mRNA isolation and analysis of in vitro translated proteins. Can J For Res 24: 2030–2033.
Halford WP, Falco VC, Gebhardt BM and Carr DJJ (1999) The inherent quantitative capacity of the reverse transcription-polymerase chain reaction. Anal Biochem 266: 181–191.
John ME (1992) An efficient method for isolation of RNA and DNA from plants containing polyphenolics. Nucleic Acids Res 20: 2381.
Jones LJ, Yue ST, Cheung C-Y and Singer VL (1998) RNA quantitation by fluorescence-based solution assay: RiboGreen reagent characterization. Anal Biochem 265: 368–374.
Lewinsohn E, Steele CL and Croteau R (1994) Simple isolation of functional RNA from woody stems of gymnosperms. Plant Mol Biol Reptr 12: 20–25.
Lewis R (1997) Kits take the trickiness out of RNA isolation, purification. The Scientist 11: 16–17.
Sambrook J, Fritsch EF and Maniatis T (1989) Molecular Cloning, A Laboratory Manual. Cold Spring Harbour Laboratory Press, Cold Spring Harbour, New York.
Schneiderbauer A, Sandermann H and Ernst D (1991) Isolation of functional RNA from plant tissues rich in phenolic compounds. Anal Biochem 197: 91–95.
Schultz DJ, Craig R, Cox-Foster DL, Mumma RO and Medford JI (1994) RNA isolation from recalcitrant plant tissue. Plant Mol Biol Reptr 12: 310–316.
Soni R and Murray JAH (1994) Isolation of intact DNA and RNA from plant tissues. Anal Biochem 218: 474–476.
Wan C-Y and Wilkins TA (1994) A modified hot borate method significantly enhances the yield of high-quality RNA from cotton (Gossypium hirsutum L.). Anal Biochem 223: 7–12.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kiefer, E., Heller, W. & Ernst, D. A simple and efficient protocol for isolation of functional RNA from plant tissues rich in secondary metabolites. Plant Mol Biol Rep 18, 33–39 (2000). https://doi.org/10.1007/BF02825291
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02825291