Summary
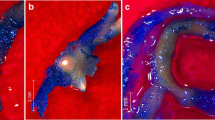
Yellowish compact callus, induced from cowpea hypocotyls on Murashige and Skoog(MS) medium (1962) containing 0.2 mg/l(0.93 μM) kinetin and 0.4 mg/l (1.81 μM) 2,4-dichlorophenoxyacetic acid (2,4-D), was subcultured on MS medium containing cytokinin alone, auxin alone, or auxins plus cytokinins in order to determine the effect of cytokinins on root organogenesis in callus cultures. The callus actively proliferated on the same medium but did not show any organogenic activity macroscopically as well as microscopically. On medium with N6-benzyladenine (BA) and 1-naphthaleneacetic acid (NAA), the yellowish compact callus first changed to pale green compact callus and then many green spots appeared on its surface under light culture. But the yellowsih compact callus remained yellowish and white spots appeared on its surface in dark culture. These spots gradually became white nodular structures. Adventitious root formation from the nodular structures occurred not only on the same medium, but also on medium with either auxin or cytokinin but not both. Yellowish compact callus on medium with auxin alone was transformed to yellowish friable callus, which did not develop adventitious roots. The yellowish friable callus could gain rhizogenic activity only after morphological modification to pale green compact callus on medium with auxin plus cytokinin. The modified callus did not form adventitious roots on medium with auxins but only with cytokinins. Therefore, it is suggested that cytokinins have stimulating effects on root formation from callus that previously did not show rhizogenic activity on medium with auxins alone. In addition, the rhizogenic potential of cowpea callus was discriminated from that of leaf explants, which formed adventitious roots directly on medium with auxin alone.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amitha, K.; Reedy, T. P. Regeneration of plantlets from different explants and callus cultures of cowpea (Vigna unguiculata L.). Phytomorphology 46:207–211; 1996.
Bae, H. H.; Cho, D.-Y.; Kim, S. G. et al. Effects of 2,4-dichlorophenoxyacetic acid on adventitious root formation from callus ofBupleurum falcatum L. and its histological observation. Korean J. Plant Tissue Cult. 22: 41–46; 1994.
Binh, D. Q.; Heszky, L. E.; Gyulai, G. et al. Plant regeneration from callus ofPuccinellia distans (L.) Parl. Plant Cell Tissue Organ Cult. 18: 195–200; 1989.
Bollmark, M.; Eliasson, L. Effects of exogenous cytokinins on root formation in pea cuttings. Physiol. Plant. 68: 662–666; 1986.
Chen, S. Y.; Read, P. E.; Hall, J. W. Influence of light, kinetin, 2,4-D and physical treatment ofRumohra adiantiformis rhizome tips culturedin vitro. Am. J. Bot. 72: 921 (abstr.); 1985.
Chin, C. K. Promotion of shoot formation inAsparagus in vitro by ancimidol. HortScience 17:590–591; 1982.
Cho, D. Y. Distribution and quantitative determination on IAA by high performance liquid chromatography on adventitious root formation in Azukia epicotyl cuttings. Korean J. Plant Tissue Cult. 12: 79–87; 1985.
Cho, D. Y.; Soh, W. Y. Effect of endogenous IAA transport on adventitious root formation inPhaseolus vulgaris hypocotyl cuttings. Korean J. Bot. 32: 323–330; 1989.
De Klerk, G.-J.; Keppel, M.; Brugge, J. T., et al. Timing of the phases in adventitious root formation in apple microcuttings. J. Exp. Bot. 46:965–972; 1995.
Drew, R. A.; Simpson, B. W.; Osborne, W. J. Degradation of exogenous indole-3-butyric acid and riboflavin and their influence of rootings response of papayain vitro. Plant Cell Tissue Organ Cult. 26:29–34; 1991.
Fabijan, D.; Taylor, J. S.; Reid, D. M. Adventitious rooting in hypocotyls of sunflower (Helianthus annuus) seedlings II. Action of gibberellins, cytokinins, auxins and ethylene. Physiol. Plant. 53:589–597; 1981.
Gautheret, R. J. Investigation on the root formation in the tissues ofHelianthus tuberosus culturedin vitro. Am. J. Bot. 56: 702–717; 1969.
Gill, R.; Eapen, S.; Rao, P. S. Tissue culture studies in mothbean-factors influencing plant regeneration from seedling explants of different cultivars. Proc. Indian Acad. Sci. 96:55–61;1986.
Gonzalez, A.; Casares, A.; Sanchez, T. R., et al. Adventitious root induction inCorylus avellana L. cotyledon slices. In Vitro Cell. Dev. Biol. 27P:125–131; 1991.
Harris, G. P.; Hart, E. M. H. Regeneration from leaf squares ofPeperomia sanderssi ADC, a relationship between rooting and budding. Ann. Bot. 28:509–591; 1964.
Heszky, L. E.; Binh, D. Q.; Kiss, E., et al. Increase of green plant regeneration efficiency by callus selection inPuccinellia limosa (Schur.) Hormbg. Plant Cell Rep. 8:174–177; 1989.
Hill, R. A.; Tuskan, G. A.; Boe, A. A.,In vitro propagation ofHosta sieboldiana using excised ovaries from immature florets. Plant Cell Tissue Organ Cult. 17:71–75; 1989.
Hussey, G. P.; Stacey, N. J.In vitro propagation of potato (Solanum tuberosum L.). Ann. Bot. 48:509–526; 1981.
Jay-Allemand, C.; De Pons, V.; Doumas, P., et al.In vitro root development from walnut cotyledons: a new model to study the rhizogenesis process in woody plants. Compte Rendue Academie des Sciences, Paris 312:369–375; 1991.
Kang, M. K.; Cho, D.-Y.; Soh, W.-Y. Effects of auxins on adventitious root formation on cotyledon-derived microcalli in lettuce (Lactuca sativa L.). Korean J. Plant Tissue Cult. 23:135–139; 1995.
Kim, S. G.; Soh, W. Y.; Cho, D. Y. Saikosaponin content in adventitious root formed from callus ofBupleurum falcatum. Korean J. Plant Tissue Cult. 22:29–33; 1995.
Ladeinde, T. A. O.; Soh, W. Y. Effect of different growth regulators on organ-ogenesis and total fresh weight gain in cultured leaf tissues of cowpea (Vigna unguiculata (L.)). Phytomorphology 41:99–107; 1991.
Letham, D. S. Cytokinins. In: Latham, D. S.; Goodwin, P. B.; Higgins, T. J. V., eds. Phytohormones and related compounds. Vol. 1. A comprehensive treatise. Amsterdam, Netherlands. Elsevier/North Holland; 1978:205–263.
McCown, B. H. Adventitious rooting of tissue cultured plants. In: Davis, T.D.; Hassig, B. E.; Sankhla, N. eds. Adventitious root formation in cuttings. Portland, Oregon: Dioscorides Press; 1988:289–302.
Meyers, J. R.; Kysely, W.; Lazzeri, P. A., et al. Protoplast isolation and culture ofGlycine species with plant regeneration ofG. canescens. Int. Congr. Plant Tissue Cell Cult. 1986; 269.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassay with tobacco tissue culture. Physiol. Plant. 15:73–93; 1962.
Nemeth, G. Benzyladenine-stimulated rooting in fruit tree stocks culturein vitro. Z. Pflanzenphysiol. 95:389–396; 1979.
Pandey, P.; Bansal, Y. K. Plantlet formation from callus cultures of cowpea (Vigna sinensis). Curr. Sci. 58:394–395; 1989.
Pierik, R. L. M..In vitro culture of higher plants. Dordrecht, Netherlands: Martinus Nijhoff Publishers; 1987:203–209.
Remeshree, A. B.; Hariharan, M.; Unnikrishnan, K. Micropropagation and callus induction ofAristolochia bracteolata Lam.—a medicinal plant. Phytomorphology 44:247–252; 1994.
Schefler, W. C. Statistics for the biological sciences. Chapter 6. Hypothesis testing. Menlo Park, California: Addison-Wesley Publ. Co.; 1980: 69–98.
Selby, C.; Kennedey, S. G.; Harvey, B. M. R. Adventitious root formation in hypocotyl cuttings ofPicea Sitchensis (Bong.) Carrthe: influence of plant growth factors. New Phytol. 120:453–457; 1992.
Sharma, V. K.; Kothari, S. L. High frequency plant regeneration in tissue cultures ofGlycine clandestina—a wild relative of soybean. Phytomorphology 43:29–33; 1993.
Van Staden, J.; Harty, A. R. Cytokinins and adventitious root formation. In: Davis, R. D.; Haissig, B. E.; Sanklha, N., eds. Adventitious root formation in cuttings. Portland, Oregon: Dioscorides Press; 1988: 185–201.
Vincent, K.; Mathew, K. M.; Hariharan, M. Micropropagation ofKaempferia galanga L., a medicinal plant. Plant Cell Tissue Organ Cult. 28:229–230; 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Soh, WY., Choi, PS. & Cho, DY. Effects of cytokinin on adventitious root formation in callus cultures ofVigna unguiculata (L.) walp. In Vitro Cell.Dev.Biol.-Plant 34, 189–195 (1998). https://doi.org/10.1007/BF02822707
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02822707