Abstract
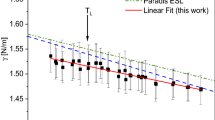
The Knudsen effusion method was combined with a mass spectrometer sensing technique in order to measure thermodynamic quantities of solid solution alloys of V-Ti binary system. The technique makes use of the regular solution model in order to treat the vaporization data in terms of ion current ratios obtained from the mass spectrometer. Within the experimental temperature range of 1500° to 1725°C and the composition range of 0.10 <N Ti<0.90, the activity of titanium in the bcc (β) phase relative to pure titanium for the binary alloy is:RT lna Ti=RT lnN Ti+N 2V (1.82±0.13) kcal per g-atom while the activity of vanadium is:RT lna V=RT lnN V+N 2Ti (1.82±0.13) kcal per g-atom.
The experimental result on the pairwise interaction parameter agrees well with the theoretically calculated value.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. R. Belton and R. J. Fruchan:J. Phys. Chem., 1967, vol. 71, p. 1403.
G. R. Belton and R. J. Fruchan:Trans. TMS-AIME, 1969, vol. 245, p. 113.
R. B. Reese, R. A. Rapp, and G. R. St. Pierre:Trans. TMS-AIME, 1968, vol. 242, p. 1719.
M. Hansen:Constitution of Binary Alloys, 2nd ed., p. 1317. McGraw Hill Book Co., New York, 1958.
J. F. Murdock and C. J. McHargue:Acta Met., 1968, vol. 16, p. 493.
L. R. Bidwell:J. Chem. Phys., 1970, vol. 52, p. 1987.
R. J. Usell and M. Hoch:Thermodynamics of Titanium Alloys II. The Titanium-Aluminum System,” Met Trans. 1971, vol. 2, pp. 2627–32.
J. V. Hackworth, M. Hoch, and H. L. Gegel:Met. Trans., 1971, vol. 2, pp. 1799–1806.
M. Hoch, R. J. Usell, J. V. Hackworth, H. L. Gegel:Proc. Intern. Conf. on Titanium, London. England. 1968. Available from M. Hoch. Univ. of Cincinnati.
R. Fowler and E. A. Guggenheim:Statistical Thermodynamics, pp. 570–82. Cambridge University Press, London, 1939.
H. K. Hardy:Acta. Met., 1953, vol. 1, p. 202.
C. H. P. Lupis and J. F. Elliott:Acta Met., 1967, vol. 15, p. 265.
W. C. Wiley and I. H. McLaren:Rev. Sci. Instr., 1955, vol. 26, p. 1150.
D. C. Damoth:Adv. Anal. Chem. Instr., vol. 4, p. 126, John Wiley and Sons, N.Y., 1962.
R. Speiser and J. W. Spretnak:Vacuum Metallurgy, p. 155, Electrochem. Soc., 1955.
P. Bolsaitis and L. Skolnick:Trans. TMS-AIME, 1968, vol. 242, p. 225.
H. S. Carslaw and J. C. Jaeger:Conduction of Heat in Solids, 2nd ed., pp. 70–72, Oxford University Press, New York, 1959.
M. Hoch: AFML TR 69-209, Air Force Materials Laboratory, Wright Patterson Air Force Base, Ohio, 1969.
Y. Beers:Introduction to the Theory of Error, 2nd ed., pp. 37–45, Addison-Wesley Publishing Company, Reading, Mass., 1957.
L. Kaufman and H. Bernstein: Tech. Rept. 14, Manlabs, Inc., Cambridge, Mass., 1968.
J. W. Ötvös and D. P. Stevenson:J. Am. Chem. Soc., 1956, vol. 78, p. 546.
J. B. Mann:J. Phys. Chem., 1967, vol. 46, p. 1646.
D. O. Van Ostenburg, D. J. Lam, H. O. Trapp, and D. F. MacLeod:Phys. Rev., 1962, vol. 128, p. 1550.
M. Hoch:Trans. TMS-AIME, 1962, vol. 224, p. 379.
R. B. Evans III, G. M. Watson, and E. A. Mason:J. Chem. Phys., 1961, vol. 35, p. 2076.
R. A. Swalin:Thermodynamics of Solids, pp. 156–63, John Wiley & Sons, N.Y., 1962.
E. J. Rolinski, C. J. Oblinger, and M. Hoch:Intern. J. Mass Spectrom. Ion Phys., 1970, vol. 5, pp. 407–414.
D. Hurley, C. J. Oblinger, and E. J. Rolinski:J. Vac. Sci. Tech., 1971, vol. 8, No. 2, pp. 461–463.
Author information
Authors and Affiliations
Additional information
EDMUND J. ROLINSKI, formerly Graduate Student, Department of Materials Science and Metallurgical Engineering, University of Cincinnati, Cincinnati, Ohio
This paper is based upon a portion of a thesis submitted by EDMUND J. ROLINSKI submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Materials Science at the University of Cincinnati.
Rights and permissions
About this article
Cite this article
Rolinski, E.J., Hoch, M. & Oblinger, C.J. Determination of thermodynamic interaction parameters in solid V-Ti alloys using the mass spectrometer. Metall Trans 2, 2613–2618 (1971). https://doi.org/10.1007/BF02814902
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02814902