Abstract
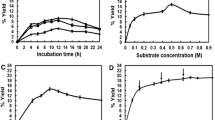
This paper describes cell-free enzymatic syntheses of sucrose and trehalose using partially-purified preparations of sucrose and trehalose synthetase. The coupling of the regeneration of uridine-5′-diphosphoglucose (UDP-Glc) with synthesis of the disaccharide offers a practical route to millimol quantities of these carbohydrates. The syntheses used pyruvate kinase, UDP-Glc pyrophosphorylase, and inorganic pyrophosphatase, and the regenerated UDP-Glc was cycled approximately 10 times.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pontis, H. G. (1977),International Review of Biochemistry, Plant Biochemistry 11, vol. 13, Northcote, D. H., ed., University Park Press, Baltimore, MD, pp. 79–117.
Birch, G. G. (1963),Advances in Carbohydrate Chemistry, vol. 18, Wolfrom, M. L., ed., Academic, New York, pp. 201–255; and Elbein, A. D. (1974),Advances in Carbohydrate Chemistry and Biochemistry, vol. 30, Tipson, R. S. and Horton, D., eds., Academic, New York, pp. 227–256.
Lemieux, R. U. and Huber, G. (1953),J. Am. Chem Soc. 75, 4118.5; and Lemieux, R. U. and Bauer, H. F. (1954),Can. J. Chem. 32, 340–344.
For a g-scale synthesis of trehalose from maltose, maltose phosphorylase, and trehalose phosphorylase,see Otsuka Food Industry Co., Ltd., Japan Kokai Tokko Koho JP 58,216,695,216,695 December 12, 1983 (1984), (Chem. Abst. 100, 173186b.)
Cardini, C. E., Leloir, L. F., and Chiriboga, C. (1955),J. Biol. Chem. 214, 149–155.
Leloir, L. F. and Cardini, C. E. (1955),J. Biol. Chem. 214, 156–165.
Cabib, E. and Leloir, L. F. (1958),J. Biol. Chem. 231, 259–276.
Nikaido, H. and Hassid, W. H. (1971),Advances in Carbohydrate Chemistry and Biochemistry, vol. 26, Tipson, R. S. and Horton, D., eds., Academic, New York, pp 351–483.
Bean, R. C. and Hassid, W. Z. (1955),J. Am. Chem. Soc. 77, 5737–5738.
Avigad, G. and Milner, Y. (1966),Methods in Enzymology, vol. 28, Neufeld, E. and Ginsburg, V., eds., Academic, New York, pp. 341–345.
Cabib, E. and Leloir, L. F. (1958),J. Biol. Chem. 231, 259–276.
Lapp, D. F., Patterson, B. W. and Elbein, A. D. (1973),Methods in Enzymology, vol. 28, Ginsberg, V., ed., Academic, New York, pp. 515–519.
Zemek, J. and Bauer, S. (1972),Biochim. Biophys. Acta 264, 393–397.
Lapp, D., Patterson, B. W. and Elbein, A. D. (1971),J. Biol. Chem. 246, 4567–4579.
Liu, C., Patterson, B. W., Lapp, D. and Elbein, A. D. (1969),J. Biol. Chem. 244, 3728–3731.
Pollak, A., Blumenfeld, H., Wax, M., Baughn, R. L. and Whitesides, G. M. (1980),J. Am. Chem. Soc. 102, 6324–6336. Pollak, A., Baughn, R. L., Adalsteinsson, O. and Whitesides, G. M. (1978), ibid.,100, 302–304.
Kayne, F. J. (1973),The Enzymes,vol. 8, 3rd ed.; Boyer, P. O., ed., Academic, New York, pp. 352–382.
Ray, W. J. and Peck, E. J. (1972),The Enzymes, vol. 6, 3rd ed.; Boyer, P. O., ed., Academic, New York, pp. 407–476.
Leloir, L. F. and Cabib, E. (1962),Methods in Enzymology; vol. 5, Colowick, S. P. and Kaplan, N. O., eds., Academic, New York, pp. 165–167.
For other examples of enzyme-catalyzed formation of glycosidic bonds on a preparative scale, see Whitesides, G. M. and Wong, C.-H. (1985),Angew. Chem. Intern. Ed. Engl. 24, 617–638, and references therein.
Nunez, H. A. and Barker, R. (1980),Biochemistry 19, 489–495.
Bednarski, M., Chenault, H. K., Simon, E. S. and Whitesides, G. M. (1987),J. Am. Chem. Soc. 109, 1283–1285.
Strominger, J. L., Maxwell, E. S., and Kalckar, H. M. (1957),Methods in Enzymology, vol. 3, Colowick, S. P. and Kaplan, N. O., eds, Academic, New York, pp. 975, 976.
Bergmeyer, H. U. and Bernt, E. (1974),Methods of Enzymatic Analysis, vol. 3, Bergmeyer, H. U. ed., Verlag-Chemie, New York, pp. 1176–1179.32).
Hey, A. E. and Elbein, A. D. (1968),J. Bacteriol. 96, 105–110.
Gawehn, K. (1974),Methods of Enzymatic Analysis, vol. 3, Bergmeyer, H. U., ed., Verlag-Chemie, New York, pp. 2239–2245.
Fiske, C. H. and Subbarow, Y. (1925),J. Biol. Chem. 66, 375–400.
Czok, R. and Lamprecht, W. (1974),Methods of Enzymatic Analysis, vol. 3, Bergmeyer, H. U., ed., Verlag-Chemie, New York, pp. 1146–1451.
Gutmann, I. and Bernt, E. (1974),Methods of Enzymatic Analysis, vol. 2, Bergmeyer, H. U., ed., Verlag-Chemie, New York, pp. 774–778.
Lang, G. and Michal, G. (1974),Methods of Enzymatic Analysis, vol. 3, Bergmeyer, H. U., eds., Verlag-Chemie, New York, pp. 1238–1242.
Bergmeyer, H. U., Bernt, E., Schmidt, F., and Stork, H. (1974),Methods of Enzymatic Analysis, vol. 3, Bergmeyer, H. U., eds., Verlag-Chemie, New York, pp. 1196–1201.
Bernt, E. and Bergmeyer, H. U. (1974),Methods of Enzymatic Analysis, vol. 3, Bergmeyer, H. U., eds., Verlag-Chemie, New York, pp. 1304–1307.
Bergmeyer, H. U. and Mical, G. (1974),Methods of Enzymatic Analysis, vol. 3, Bergmeyer, H. U., eds., Verlag-Chemie, New York, pp. 1233–1237.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951),J. Biol. Chem. 193, 265–275.
Schwarzenbach, P. (1976),J. Chromatography 117, 206–210; Meagher, R. B. and Furst, A. (1976),J. Chromatography 117, 211–215; Brandao, S. C., Richmond, M. L., Gray, J. L., Morton, I. D., and Stine, C. M. (1980),J. Food Sci. 45, 1492, 1493.
The Merck Index, 10th ed. (1983), Windholz, M., eds., Merck, Rahway, NJ, p. 1273.
Panek, A. D. (1969),J. Bacteriology 99, 904, 905.
Kelly, P. J. and Catley, B. (1976),J. Analytical Biochem. 72, 353–358.
The Merck Index, 10th ed. (1983), Windholz, M., ed., Merck, Rahway, NJ, p. 1370.
Author information
Authors and Affiliations
Additional information
This work was supported by the NIH through several grants, most recently GM 30367.
Rights and permissions
About this article
Cite this article
Haynie, S.L., Whitesides, G.M. Enzyme-catalyzed organic synthesis of sucrose and trehalose with in situ regeneration of UDP-Glucose. Appl Biochem Biotechnol 23, 155–170 (1990). https://doi.org/10.1007/BF02798384
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02798384