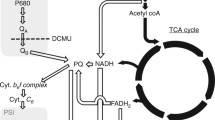
Abstract
We have developed a photochemical cell using a combination of photosynthetic electron transport (photosystem I particles) and the photoreduction of a dye such as flavin mononucleotide (FMN) (6). The overall power conversion efficiency depends on the rate of charge transfer across the electrode surfaces in addition to the efficiency of the photosynthetic and photochemical reactions. For this reason, we studied the effect of varying the nature of the electrodes on the power developed. We found that reticulated vitreous carbon electrodes showed higher power conversion efficiencies than did nickel mesh, platinum, or SnO2 glass. There are two reasons for this. First, the ratio of actual to apparent surface area is greater for RVC electrodes than for the others. Second, FMN and its photoproducts react better with carbon than platinum electrodes. Substituting RVC electrodes for platinum increased the power conversion efficiency from 1.0 to 3.9%. Platinizing platinum, nickel mesh, or brass electrodes also increased the power developed. However, the photopotential remained stable for several hours only for the platinized platinum electrodes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- chl:
-
chlorophyll
- DCIP:
-
dichlorophenol indophenol
- EDTA:
-
ethylendiamine tetraacetic acid
- FMN:
-
flavin mononucleotide
- PSI:
-
Photosystem I
- Pt:
-
platinum
- PtzPt:
-
platinized platinum
- RVC:
-
reticulated vitreous carbon
- σ:
-
electrode current density
- TCA:
-
trichloroacetic acid
- Tricine:
-
N-tris(hydroxymethyl) methyl glycine
References
Gross, E. L., Youngman, D. R., and Winemiller, S. L. (1978),Photochem. Photobiol. 28, 249.
Bhardwaj, R., Pan, R. L., and Gross, E. L. (1981),Nature 289, 396.
Bhardwaj, R., Pan, R. L., and Gross, E. L. (1981),Photochem. Photobiol. 34, 215.
Bhardwaj, R., Pan, R. L., and Gross, E. L. (1981), inProc. Fifth International Phososysnthesis Congress, vol. 6, Akoyunoglou, G., ed., Balaban International Science Services, Philadelphia, Pa., pp. 719–728.
Bhardwaj, R., Pan, R. L., and Gross, E. L. (1981),Photobiochem. Photobiophys. 3, 19.
Pan, R. L., Fan, I.-J., Bhardwaj, R., and Gross, E. L. (1982),Photochem. Photobiol. 35, 655.
Shiozawa, J. A., Alberte, R. S., and Thornber, J. P. (1974),Arch. Biochem. Biophys. 1651, 388.
Tien, H. T. (1974), inBilayer Lipid Membranes (BLM): Theory and Practice, Dekker, NY.
Haehnel, W., Heupel, A., and Hengstermann, D. (1978),Z. Naturforsch. 336c, 392.
Gross, E. L., and Grenier, J. (1978),Arch. Biochem. Biophys. 187, 387.
Arnon, D. I. (1949),Plant Physiol. 24, 1.
Hemmerich, P., Veeger, C., and Wood, H. C. S. (1965),Angew. Chem. Int. Ed.4, 671.
Gross, E. L., and Sanderson, D. G., unpublished data.
Allen, M. J. (1977), inLiving Systems as Energy Converters, Buvet, R., Allen, M. J., and Massue, J. P., eds, Elsevier North Holland, Amsterdam, pp. 271–274.
Haehnel, W., and Hochheimer, H. J. (1979),J. Electroanal. Chem. 104, 563–574.
Bulletin No. 176, Chemtronics International, Inc., Ann Arbor, MI.
Norvell, V. E., and Mamantov, G. (1977),Anal. Chem. 49, 1470.
Strohl, A. N., and Curran, P. J. (1979),Anal. Chem. 51, 353.
Strohl, A. N., and Curran, P. J. (1979),Anal. Chem. 51, 1045.
Strohl, A. N., and Curran, P. J. (1979),Anal. Chem. 51, 1050.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sanderson, D.G., Pan, R.L. & Gross, E.L. Studies of a photosynthetic photoelectrochemical cell using various electrodes. Appl Biochem Biotechnol 8, 395–405 (1983). https://doi.org/10.1007/BF02779912
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02779912