Summary
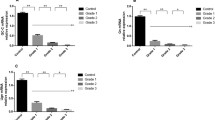
Experimental ulcerative colitis was produced in rats and the changes of PGE2 levels in the colonic mucosa and the tissue localization of PGE2 were studied immunohistochemically during the process of onset and healing of the experimental lesions. Compared with that in controls, PGE2 levels in colonie mucosa were not significantly higher in the early stage of inflammation, but rose gradually with exacerbation of inflammation, and after reaching peak values at the peak stage of development of colitis, decreased as signs of inflammation receded. Immunohistochemical study of the normal rat colon showed that PGE2 was uniformly positive in the muscularis mucosa and in the tunica propria muscularis adjacent to the submucosal layer. Furthermore, as the inflammation progressed infiltrating inflammatory cells also became positive, especially macrophages. These changes decreased along with regeneration of the epithelium and resolution of the inflammation. Glandular epithelial cells did not show positivity. These results made it clear that the PGE2 level in colonie mucosa is an useful index of the activity of colitis and that changes in PGE2 levels are associated with infiltration by inflammatory cells, principally macrophages, of the interstitium. The results also suggested the possibility that the muscularis mucosa plays a role in the colonie mucosal protective system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ramptom DS, Hawkey CJ. Prostaglandins and ulcerative colitis. Gut 1984;25:1399–1413.
Sharon P, Ligumsky M, Rachmilewitz D, et al. Role of prostaglan dins in ulcerative colitis: enhanced production during active dis ease and inhibition by sulfasalazine. Gastroenterology 1978;75: 638–640.
Ramptom DS, Sladen GE, Youlten LJF. Rectal mucosal prosta glandin E2 release and its relation to disease activity, electrical potential difference, and treatment in ulcerative colitis. Gut 1980;21:591–596.
Yamazaki H, Hiwatashi N, Goto Y. Arachidonic acid metabolism in inflammatory bowel disease: with reference to cyclooxygenase products. Jpn J Gastroenterol 1985;82:2562–2568. (in Japanese)
Ohkusa T. Production of experimental ulcerative colitis in ham sters by dextran sulfate sodium and change in intestinal micro flora. Jpn J Gastroenterol1985;82:1327–1336. (in Japanese)
Arakawa T, Satoh H, Fukuda T, et al. Orcadian rhythm of prosta glandin system in rat stomach. Jpn J Gastroenterol 1988;85:655–658. (in Japanese)
Arakawa T, Nakamura H, Chono S, et al. Prostaglandin E2 in the rat gastric mucosa (First report): establishment of assay procedure and effects of non-steroidal anti-inflammatory compounds. Jpn J Gastroenterol1980;77:1052–1059. (in Japanese)
Matts SGF. The value of rectal biopsy in the diagnosis of ulcerative colitis. Quart J Med 1961;30:393–407.
Matsumoto T, Higuchi K, Arakawa T, et al. Immunohistochemi cal localization of prostaglandin E2 (PGE2) in rat gastric mucosa. Gastroenterology 1987;92:1522. (abstract)
Saito H, Sato J, Mizuno S, et al. Immunohistochemical localiza tion of PGE2 and 6-keto PGF1 in gastric mucosa of chronic gastri tis. Therapeutic Research 1990;ll:1243–1249. (in Japanese)
Horton EW, Main IHM, Thompson CJ, et al. Effect of orally admin istered prostaglandin E1 on gastric secretion and gastrointestinal motility in man. Gut 1968;9:655–658.
Gould SR. Prostaglandins, ulcerative colitis and sulphasalazine. Lancet 1975;2:988.
Sharon P, Stenson WF. Metabolism of arachidonic acid in acetic acid colitis in rats: similarity to human inflammatory bowel dis ease. Gastroenterology 1985;88:55–63.
Zipser RD, Nast CC, Lee M, et al. In vivo production of leuko triene B4 and leukotriene C4 in rabbit colitis: relationship to inflammation. Gastroenterology 1987;92:33–39.
Hiramatsu M, Sakai M, Uchino H, et al. Cecal and colonie blood flow in the early developing stage of rat experimental ulcerative colitis induced by dextran sulfate sodium. Gastroenterol Endosc 1988;30:2567–2577. (in Japanese)
Rampton DS, Sladen GE. Prostaglandin synthesis inhibitors in ulcerative colitis: flurbiprofen compared with conventional treatment. Prostaglandins 1981;21:417–425.
Perez G, Mcguckin J. Cellular localization of prostaglandin A2 in the rat kidney. Prostaglandins 1972;2:393–398.
Ogra SS, Kirton KT, Tomasi TB, et al. Prostaglandins in the human fallopian tube. Fertility and Sterility 1974;25:250–255.
Husby G, Stricland RG, Rigler GL, et al. Direct immunochemical detection of prostaglandin-E and cyclic nucleotides in human malignant tumors. Cancer 1977;40:1629–1642.
Sakata H, Yoshida H. Cellular localization of prostaglandin E in the rabbit iris by indirect immunofluorescence method. Albrecht V Grafes Arch Klin Exp Opthal1979;211:221–227.
Nakamura M, Oda M, Nishizaki Y, et al. The structure and regula tion of the muscularis mucosae of the rat stomach: histochemical and radioautographic studies. Therapeutic Research 1989;10: 3155–3166. (in Japanese)
Robert A, Negamis JE, Lancaster C, et al. Prevention, through cytoprotection, of clindamycin-induced colitis in hamsters with 16, 16-dimethyl PGE2. Gastroenterology1980;78:1245.
Wallace JL, Whittle BJR, Boughton-Smith NK. Prostaglandin pro tection of rat colonie mucosa from damage induced by ethanol. Dig Dis Sci1985;30:866–876.
Humes JL, Bonney RJ, Pelus L, et al. Macrophages synthesize and release prostaglandins in response to inflammatory stimuli. Nature 1977;269:149–151.
Katayama S, Ishii J. Physiological roles of primary prostaglandins. Journal of Clinical and Experimental Medicine 1987;143:281–286. (in Japanese)
Zifroni A, Treves AJ, Sachar DB, et al. Prostanoid synthesis by cul tured intestinal epithelial and mononuclear cells in inflammatory bowel disease. Gut 1983;24:659–664.
Eliakim R, Karmeli F, Razin E, et al. Role of platelet-activating factor in ulcerative colitis: enhanced production during active dis ease and inhibition by sulfasalazine and predonisolone. Gastroen terology 1988;95:1167–1172.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yamashita, S. Studies on changes of colonic mucosal PGE2 levels and tissue localization in experimental colitis. Gastroenterol Jpn 28, 224–235 (1993). https://doi.org/10.1007/BF02779224
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02779224