Abstract
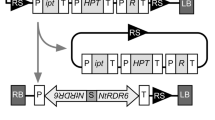
A hairpin RNA (hpRNA) vector, pKNOCKOUT (pKO) has been constructed to facilitate the analysis of posttranscriptional gene silencing (PTGS) in anAgrobacterium-mediated transient expression system developed for tobacco. The pKO binary vector was tested by cloning a firefly luciferase (Photinus pyralis) gene segment in sense (sFLUC), antisense (aFLUC), and inverted repeat (ihpFLUC) orientations. The inverted repeats of the target gene are separated by the castor bean catalase intron (CbCi) and, when transcribed and spliced, produce a self-complementary hpRNA. hpRNA-mediated gene silencing exploits a cellular mechanism that recognizes double-stranded RNA (dsRNA) and subjects it, and homologous mRNA molecules, to sequence-specific degradation. Agrobacteria harboring compatible plasmids, pE1778-RiLUC (Renilla luciferase normalization construct) and pTCaMV35S-FiLUC (functional firefly luciferase test construct), were co-infused with different variants of pKO-FLUC plasmids intoNicotiana benthamiana andNicotiana tobaccum leaf tissues. Reduced firefly luciferase reporter gene activity (from pTCaMV35S-FiLUC) indicated gene silencing and was observed in leaf tissues co-infused with pKO-sFLUC (50% reduction), pKO-aFLUC (85% reduction), or pKO-ihpFLUC (96% reduction) agrobacteria lines. Gene silencing was observed at different times postinfusion in leaves from bothNicotiana species. The hpRNA-mediated interference with FiLUC reporter gene expression, as measured by reduced firefly luciferase activity, was found to also suppress silencing of the cotransferredRenilla reniformis luciferase normalization reporter gene, resulting in reproducibly elevated RiLUC activity. This suppression effect was reduced by lowering the percentage of infused pKO-ihpFLUC agrobacteria without markedly effecting hpRNA-mediated gene silencing of FiLUC (all pKO-ihpFLUC dilutions produced >90% reduction of FiLUC activity). Viral suppressors of PTGS, such as p19 and HcPro, were found to reduce the RNAi effect of hpRNA-mediated gene silencing. To our knowledge, this is the first demonstration that excess amounts of targeted dsRNA can result in nonspecific suppression of PTGS of an unrelated, co-infused reporter gene in plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CAM:
-
coelenterazine assay media (sea pansy)
- CaMV35S:
-
cauliflower mosaic virus 35S transcript
- CbCi:
-
castor bean catalase intron
- dsRNA:
-
double-stranded RNA
- dpi:
-
days postinoculation
- DTT:
-
dithiothreitol
- GUS:
-
β-glucuronidase
- hpRNA:
-
hairpin RNA
- ihpRNA:
-
intron hairpin RNA
- Km:
-
kanamycin
- LB:
-
Luria-Bertani
- FLUC:
-
Photinus pyralis (firefly) luciferase gene segment
- FiLUC:
-
Photinus pyralis (firefly) intron-modified luciferase
- LAM:
-
luciferin assay media (firefly)
- miRNA:
-
microRNA
- Nost :
-
nopaline synthase terminator
- pBS:
-
pBluescript II (SK+)
- PTGS:
-
posttranscriptional gene silencing
- RiLUC:
-
Renilla reniformis (sea pansy) intron-modified luciferase
- RLUC:
-
Renilla reniformis (sea pansy) luciferase
- RNAi:
-
RNA interference
- RLU:
-
relative light unit
- siRNA:
-
small interfering RNA
- T-DNA:
-
transferred DNA
References
Akama K, Shiraishi H, Ohta S, Nakamura K, Okada K, and Shimura Y. (1992) Efficient transformation ofArabidopsis thaliana: comparison of the efficiencies with various organs, plant ecotypes andAgrobacterium strains. Plant Cell Reports 12: 7–11.
Akashi H, Miyagishi M, and Taira K (2001) Suppression of gene expression by RNA interference in cultured plant cells. Antisense Nucleic Acid Drug Dev 11: 359–367.
Akashi H, Miyagishi M, and Taira K (2004) RNAi expression vectors in plant cells. Methods Mol Biol 252: 533–543.
Bartel B and Bartel DP (2003) MicroRNAs: at the root of plant development? Plant Physiol 132: 709–717.
Baulcombe DC (1996) RNA as a target and an initiator of post-transcriptional gene silencing in transgenic plants. Plant Mol Biol 32: 79–88.
Becker D, Kemper E, Schell J, and Masterson R (1992) New plant binary vectors with selectable markers located proximal to the left T-DNA border. Plant Mol Biol 20: 1195–1197.
Benfey P and Chua N (1990) The cauliflower mosaic virus 35s promoter: combinatorial regulation of transcription in plants. Science 250: 959–966.
Brigneti G, Voinnet O, Li W, Ji L, Ding S, and Baulcombe D (1998) Viral pathogenicity determinants are suppressors of transgene silencing inNicotiana benthamiana. EMBO Journal 17: 6739–6746.
Cazzonelli C and Velten J (2003) Construction and testing of an intron-containing luciferase reporter gene fromRenilla reniformis. Plant Mol Biol Rep 21: 271–280.
Depicker A and Montagu MV (1997) Post-transcriptional gene silencing in plants. Curr Opin Cell Biol 9: 373–82.
Hajdukiewicz P, Svab Z, and Maliga P (1994) The small, versatile pPZP family ofAgrobacterium binary vectors for plant transformation. Plant Mol Biol 25: 989–994.
Johansen L and Carrington J (2001) Silencing on the spot. Induction and suppression of RNA silencing in theAgrobacterium-mediated transient expression system. Plant Physiol 126: 930–938.
Maiti IB and Shepherd RJ (1998) Isolation and expression analysis of peanut chlorotic streak caulimovirus (PC1SV) full-length transcript (FLt) promoter in transgenic plants. Biochem Biophys Res Commun 244: 440–444.
Mankin S, Allen G, and Thompson W (1997) Introduction of a plant intron into the luciferase gene ofPhotinus pyralis. Plant Mol Biol Rep 15: 186–196.
Ni M, Cui D, Einstein J, Narasimhulu S, Vergara Q, and Gelvin S (1995) Strength and tissue specificity of chimeric promoters derived from the octopine and mannopine synthase genes. Plant J. 7: 661–676.
Ohta S, Mita S, Hattori T, and Nakamura K (1990) Construction and expression in tobacco of a beta-glucuronidase (GUS) reporter gene containing an intron within the coding sequence. Plant Cell Physiol 31: 805–814.
Pickford AS and Cogoni C (2003) RNA-mediated gene silencing. Cell Mol Life Sci 60: 871–882.
Reynolds A, Leake D, Boese Q, Scaringe S, Marshall W, and Khvorova A (2004) Rational siRNA design and RNA interference. Nat Biotechnol 22: 326–330.
Sambrook J and Russell D (2001) Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, New York.
Scattat M, Klosgen R, and Marques J (2004) A novel vector for efficient gene silencing in plants. Plant Mol Biol Rep 22: 145–153.
Smith NA, Singh SP, Wang MB, Stoutjesdijk PA, Green AG, and Waterhouse PM (2000) Total silencing by intron-spliced hairpin RNAs. Nature 407: 319–320.
Susi P, Hohkuri M, Wahlroos T, and Kilby NJ (2004) Characteristics of RNA silencing in plants: similarities and differences across kingdoms. Plant Mol Biol 54: 157–174.
Van der Hoorn RA, Laurent F, Roth R, and De Wit PJ (2000) Agroinfiltration is a versatile tool that facilitates comparative analyses of Avr9/Cf-9-induced and Avr4/Cf-4-induced necrosis. Mol Plant Microbe Interact 13: 439–446.
Van Houdt H, Van Montagu M, and Depicker A (2000) Both sense and antisense RNAs are targets for the sense transgene-induced posttranscriptional silencing mechanism. Mol Gen Genet 263: 995–1002.
Vancanneyt G, Schmidt R, O'Connor-Sanchez A, Willmitzer L, and Rocha-Sosa M (1990) Construction of an intron-containing marker gene: splicing of the intron in transgenic plants and its use in monitoring early events inAgrobacterium-mediated plant transformation. Mol Gen Genet 220: 245–250.
Voinnet O (2001) RNA silencing as a plant immune system against viruses. Trends Genet 17: 449–459.
Voinnet O, Pinto Y, and Baulcombe D (1999) Suppression of gene silencing: A general strategy used by diverse DNA and RNA viruses of plants. PNAS 96: 14147–14152.
Voinnet O, Rivas S, Mestre P, and Baulcombe D (2003) An enhanced transient expression system in plants based on suppression of gene silencing by the p19 protein in tomato bushy stunt virus. Plant J 33: 949–956.
Walkerpeach C and Velten J (1994)Agrobacterium-mediated gene transfer to plant cells cointegrate and binary vector systems. In: Gelvin S, Schilperoort R (eds) Plant Molecular Biology Manual, pp. 1–19. Kluwer Academic Publishers, Belgium.
Waterhouse PM, Graham MW, and Wang MB (1998) Virus resistance and gene silencing in plants can be induced by simultaneous expression of sense and antisense RNA. Proc Natl Acad Sci USA 95: 13959–13964.
Wesley SV, Helliwell CA, Smith NA, Wang MB, Rouse DT, Liu Q, Gooding PS, Singh SP, Abbott D, Stoutjesdijk PA, and others (2001) Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J 27: 581–590.
Wesley SV, Liu Q, Wielopolska A, Ellacott G, Smith N, Singh S, and Helliwell C (2003) Custom knock-outs with hairpin RNA-mediated gene silencing. Methods Mol Biol 236: 273–286.
Xie Z, Kasschau KD, and Carrington JC (2003) Negative feedback regulation of Dicer-Like1 inArabidopsis by microRNA-guided mRNA degradation. Curr Biol 13: 784–789.
Author information
Authors and Affiliations
Additional information
Disclaimer: Mention of commercial or proprietary products does not constitute an endorsement by the USDA. The USDA offers its programs to all eligible persons regardless of race, color, age, sex, or national origin.
Rights and permissions
About this article
Cite this article
Cazzonelli, C.I., Velten, J. Analysis of RNA-mediated gene silencing using a new vector (pKNOCKOUT) and an in plantaAgrobacterium transient expression system. Plant Mol Biol Rep 22, 347–359 (2004). https://doi.org/10.1007/BF02772678
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02772678