Abstract
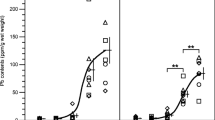
Analysis and distribution of Pb and Cd in different mice organs, including the liver, kidney, spleen, heart, and blood, were evaluated before and after treatment with different aqueous concentrations of Nigella sativa (1.25–10.0 mg/L). Atomic absorption spectrometry was used for analysis of Pb and Cd in these organs. Results indicated that the Pb in the unexposed group of mice without treatment with N. sativa (black cumin) was in the following order: liver>heart>spleen>kidney, and the distribution of Pb in various organs of the unexposed group was not affected significantly by N. sativa. Moreover, results of mice exposed for Pb show that the Pb concentrations in different organs were reduced significantly (p<0.05) by 72.9%, 63.4%, 72.3%, 66.7%, and 39.5% at a dose of 10 mg/L of N. sativa for the liver, kidney, heart, spleen, and blood, respectively. Furthermore, the distribution of Cd in the unexposed Cd group of mice without treatment with N. sativa was in the following order: kidney>heart>spleen>liver. Nigella sativa at 10 mg/L reduced Cd levels in mice exposed to Cd by 75.5%, 83.3%, 47.0%, 95.3%, and 100% in the liver, kidney, heart, spleen, and blood, respectively, whereas blood Cd concentrations were lowered to below the detection limit of 0.05 μg/L.
A 28-d exposure of mice to a Cd−Pb mixture at a concentration of 1 ppm in drinking water induced a highly significant inhibition (p<0.0001) of antibody response to human serum (80.5%). The suppressed immune responses in mice pretreated with the Cd−Pb mixture were reversed by 43.1% and 38.9% in the presence of 1.25 and 2.5 mg/mL of N. sativa, respectively, whereas higher concentrations (5–10 mg/mL) of N. sativa increased the immunosuppression significantly. Nigella sativa at 1.25–10 mg/mL did not induce any significant modulation of the antibody response in unexposed mice.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
World Health Organization, Evaluation of Criteria Food Additives and Contaminants, Technical Report Series No. 837, WHO, Geneva (1993).
World Health Organization, Environmental Health Criteria 165, International Programme on Chemical Safety, WHO, Geneva (1995).
E. Silbergeld, The Elimination of Lead from Gasoline: Impacts of Lead in Gasoline on Human Health, and the Costs and Benefits of Eliminating Lead Additives, The World bank, Washington, DC (1996).
R. A. Goyer, Results of lead research: prenatal exposure and neurological consequences, Environ. Health Perspect., 104(10), 1050–1054 (1996).
National Research Council, Measuring Lead Exposure in Infants, Children and Other Sensitive Populations, National Academy Press, Washington, DC (1993).
M. G. Cherian, and R. A. Goyer. Cadmium toxicity. Commun. Toxicol. 3, 191–206 (1989).
S. Benoff, A. Jacob, and I. R. Hurley. Male infertility and environmental exposure to lead and cadmium. Hum. Rep. Update 6, 107–121 (2000).
J. Ye, S. Wang, M. Barger, V. Castranova, and X. Shi, Activation of androgen response element by cadmium: a potential mechanism for a carcinogenic effect of cadmium in the prostate. J. Environ. Pathol. Toxicol. Oncol. 19, 275–280 (2000).
A. M. Massadeh, M. Tahat, Q. Jaradat, and I. F. Al-Momani, Lead and cadmium contamination in roadside soil in Irbid City, Jordan. J. Soil Sediment Contam: Int. J. 13(4), 347–359 (2004).
A. M. Massadeh Distribution of copper and zinc in different fractions of particle sizes in road dust samples in Irbid City, Jordan using atomic absorption spectrometry. Res. J. Chem. Environ. 7(4), 49–54 (2003).
A. M. Massadeh and R. D. Snook, Determination of Pb and Cd in road dusts over the period in which Pb was removed from petrol in the UK. J. Environ. Monitl., 4, 567–572 (2002).
A. M. Massadeh, F. Q. Alali, and Q. Jaradat, Determination of copper and zinc in different brands of cigarettes in Jordan. Acta Chim. Slov. 50, 375–381 (2003).
A. M. Massadeh, F. Q. Alali, and Q. Jaradat, Determination of cadmium and lead in different brands of cigarettes in Jordan. Environ. Monit. Assess. 104, 163–170 (2005).
A. M. Massadeh and Saafan Al-Safi, Analysis of cadmium, and lead: their immunosuppressive effects and distribution in various organs of mice. Biol. Trace Element Res. 108(1–3), 278–286 (2005).
P. E. Bigazzi, Metal and kidney autoimmunity. Environ. Health Perspect. 107, 753–765 (1999).
A. Karrkaya, B. Yccesoy, and O. S. Sardas, An immunological study on workers occupationally exposed to cadmium. Hum. Exp. Toxicol. 13, 73–75 (1994).
F. Momani and A. M. Massadeh. The Effect of different heavy metal concentrations on Drosophila melanogaster larval growth and development. Biol Trace Element Res. 108(1–3), 271–278 (2005).
A. M. Massadeh, F. Momani, and H. Haddad. Removal of lead and cadmium by hahophilic bacteria isolated from the Dead Sea shore, Jordan. Biol. Trace Element Res. 108(1–3), 259–278 (2005).
A. Abdel-Fattah, K. Matsumoto, and H. Watanabe. Antinociceptive effects of Nigella sativa oil and its major component, thymoquinone, in mice. Eur. J. Pharmacol. 400, 89–97 (2000).
M. El-Dakhakhny, Studies on the Egyptian Nigella sativa L part IV: some pharmacological properties of the seed’s active principle in comparison to its dihydro compound and its polymer. Arzneim-Forsch. 15, 1227–1229 (1965).
K. Al-Tahir, and M. Ashour, M. Al-Harbi, The cardiovascular actions of the volatile of the black seed (Nigella sativa) in rats, elucidation of the mechanism of action, Gen. Pharmacol. 24, 1123–1131 (1993).
A. A. Al-Hader, M. B. Aqel, and Z. A. Hasan, Hypoglycemic effects of the volatile oil of Nigella sativa seeds, Int. J. Pharmacogn, 31, 96–100 (1993).
M. S. M. Hanafy and M. E. Hatem. Studies on the antimicrobial activity of Nigella sativa seed (black cumin), J. Ethnopharmacol. 34, 275–278 (1991).
D. R. Worthen, A. O. Ghosheh, and P. A. Crooks, The in vitro antitumor activity of some crude and purified components of blackseed, Nigella sativa L., Anticancer Res. 18, 1527–1532 (1998).
S. M. K. Swamy and B. K. H. Tan, Cytotoxic and immunopotentiating effects of ethanolic extract of Nigella sativa L. seeds, J Ethnopharmacol. 70, 1–7 (2000).
M. S. Atta-ur-Rahman and M. K. Zaman, Nigellimine: a new isoquinoline alkaloid from the seeds of Nigella sativa, J. Nat. Prod. 55, 676–678 (1992).
F. C. Hay, Immunological manipulations in vivo, in Practical Immunology, O. Westwood, ed. Blackwell Science, Oxford (2002).
C. Taupeau, J. Poupon, F. Nome, and B. Lefevre, Lead accumulation in the mouse ovary after treatment-induced follicular atresia, Report Toxicol. 15, 385–391 (2001).
Y. Liu, J. Liu, and C. D. Klaassen, Metallothionein-null and wild-type mice show similar cadmium absorption and tissue distribution following oral cadmium, Administration, Toxicol. Appl. Pharmacol. 175, 253–259 (2001).
M. Radike, D. Warshawsky, J. Caruso, et al., Distribution and accumulation of a mixture of arsenic, cadmium, chromium, nickel, and vanadium in mouse small ntestine, kidneys, pancreas, and femur following oral administration in water or feed, J. Toxicol. Environ. Health 65, 2029–2052 (2002).
A. El-Kadi and O. Kandil, Effect of Nigella sativa (the black seeds) on immunity. Proceedings of the Fourth International Conference on Islamic Medicine, Bull Islamic Med. 4, 344–348 (1986).
A. Haq, M. Abdullatif, P. I. Lobo, K. S. A. Khabar, K. V. Sheth, and S. T. Al-Sedairy, Nigella sativa: effect on human lymphocytes and polymorphonuclear leukocyte phagocytic activity. Immunopharmacology 30, 147–155 (1995).
A. Haq, P. Lobo, M. Al-Tufail, N. Rama, and S. T. Al-Sedairy, Immunomodulatory effect of Nigella sativa proteins fractionated by ion exchange chromatography, Int. J Immunopharmacol. 21, 283–295 (1999).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Massadeh, A.M., Al-Safi, S.A., Momani, I.F. et al. Analysis of cadmium and lead in mice organs. Biol Trace Elem Res 115, 157–167 (2007). https://doi.org/10.1007/BF02686027
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02686027