Abstract
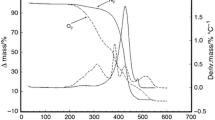
Isothermal crystallization of plam oil was studied by means of differential scanning calorimetry (DSC) as well as by nuclear magnetic resonance spectrometry to monitor its solid fat content (SFC). The temperature of crystallization (Tc) varied from 0 to 30°C, depending on the method used. The plot of %SFC vs. time at 25°C was sigmoidal in shape. However, at lower temperatures, two consecutive curves were clearly visible. Results from DSC experiments showed the following interesting features. At each Tc, the crystals produced were of different compositions. From 0 to 8°C, the thermogram showed three peaks, with the first two peaks (I and II) sharp, and the third (III) rather broad. At elevated temperatures up to 20°C, peak II disappeared totally while peak III tended to shift toward peak I. Above 20°C, both peaks shifted downward to longer times. Peak I continued to be broadened, and then suddenly disappeared at Tc above 24°C. The melting thermograms of the crystals obtained above and below this cut-off point were distinctly different. Kinetic studies on isothermal crystallization based on the data of SFC measurements showed that the data fit well into the Avrami-Erofeev equation with n=3 over the first 70% of the crystallization.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ng, W. L.,J. Am. Oil Chem. Soc. 76:879 (1990).
Persmark, U., K.A. Melin and P.O. Stahl,Rev. Ital. Sos. Gra. LIII:301 (1976).
Jacobsberg, B., and C.H. Oh,J. Am. Oil Chem. Soc. 53:609 (1976).
Kawamura, K., ——Ibid.56:753 (1979).
Kawamura, K., ——Ibid.57:48 (1980).
van Putte, K.P.A.M., and B.H. Bakker, ——Ibid.64:1138 (1987).
Yap, P.H., J.M. deMan and L. deMan,Fat Sci. Technol. 91:178 (1989).
Oh, C.H., and K.G. Berger, inPalm Oil Product Technology in the Eighties, Incorporated Society of Planters, Kuala Lumpur, 1983, p. 383.
Ng, W.L.,J. Am. Oil Chem. Soc. 66:1103 (1989).
Mullin, J.W., inCrystallisation, Butterworths, London, 1972, Chapter 5
Strickland-Constable, R.F., inKinetics and Mechanism of Crystallisation, Academic Press, London, 1968, p. 95.
Avrami, M.J. Chem. Phys. 8:212 (1940).
Ng, W.L.,Aust. J. Chem. 28:1169 (1975).
Author information
Authors and Affiliations
About this article
Cite this article
Ng, W.L., Oh, C.H. A kinetic study on isothermal crystallization of palm oil by solid fat content measurements. J Am Oil Chem Soc 71, 1135–1139 (1994). https://doi.org/10.1007/BF02675908
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02675908