Abstract
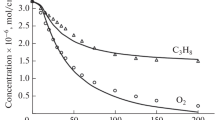
The pyrolysis and incomplete oxidation of hydrocarbons in the presence of water at high pressures is modeled numerically. The calculations show that the nonidealness of the gas can have a significant effect on the composition and temperature of the reaction products. The results of equilibrium and global kinetics calculations are in better agreement with one another, but a model with detailed kinetics fits the exprimental data better.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
V. M. Glazov and L. M. Pavlova,Chemical Thermodynamics and Phase Equilibria [in Russian], Metallurgiya, Moscow (1988).
R. C. Reid, J. M. Prausnutz, and T. Sherwood,The Properties of Gases and Liquids, McGraw-Hill, New York (1977).
C. K. Westbrook and F. L. Dryer, “Chemical kinetic modeling of hydrocarbon combustion,“ in: Prog. Engergy combust. sci,10, 1–57 (1984).
A. F. Dregalin, I. A. Zenukov, V. G. Kryukov, and V. I. Naumov,Mathematical Modeling of High-Temperature Processes in Power Plants [in Russian], Izd. Kazan’ Univ., Kazan’ (1985).
E. A. Moelwyn-Hughes,The Chemical Statics and Kinetics of Solution, Academic Press, London-New York (1971).
D. Depeyre and C. Flicoteaux, “Modeling of thermal stream cracking ofn-hexadecane,”Ind. Eng. Chem. Res.,30, 1116–1130 (1991).
H. R. Holgate, P. A. Webley, and J. W. Tester, “Carbon monoxide oxidation in supercritical water: The effects of heat transfer and water-gas shift reaction on observed kinetics,”Energy Fuels,6, 586–597 (1992).
Yu. M. Zhorov,Kinetics of Industrial Organic Reactions [in Russian], Khimiya, Moscow (1989).
Yu. M. Zhorov, I. I. Vasil’eva, G. M. Panchenkov, and S. T. Kuz’min, “Mathematical description of the pyrolysis of benzenes,”Khim. Tekhnol. Topliv Masel, No. 4, 16–19 (1975).
B. M. Fabuss, R. Kafesjian, J. O. Smith, and Ch. N. Satterfield, “Thermal decomposition rates of saturated cyclic hydrocarbon,”Ind. Eng. Chem.,3, No. 3, 132–138 (1964).
H. J. Hepp and F. E. Frey, “Pyrolysis of propane and butanes at elevated pressure,”Ind. Eng. Chem.,45, 410 (1953).
N. A. Vatolin, G. K. Moiseev, and B. G. Trusov,Thermodynamic Modeling in High-Temperature Inorganic Systems [in Russian], Metallurgiya, Moscow (1994).
R. G. Mallinson, R. L. Braun, C. K. Westbrook, and A. K. Burnham, “Detailed chemical kinetics study of the role of pressure in butane pyrolysis,”Ind. Eng. Chem. Res.,31, 37–45 (1992).
J. W. Tester, P. A. Webley, and H. R. Holgate, “Revised global kinetic measurements of methanol oxidation in supercritical water,”Ind. Eng. Chem. Res.,32, 236–239 (1993).
Author information
Authors and Affiliations
Additional information
Translated fromFizika Goreniya i Vzryva, Vol. 35, No. 3, pp. 20–28, May–June 1999.
Rights and permissions
About this article
Cite this article
Dvornikov, N.A. Equilibrium and kinetic modeling of high-pressure pyrolysis and oxidation of hydrocarbons. Combust Explos Shock Waves 35, 230–238 (1999). https://doi.org/10.1007/BF02674443
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02674443