Abstract
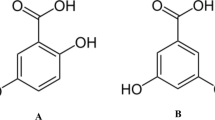
The effects of antioxidants were studied from the viewpoints of (a) the products generated during the autoxidation process and (b) the functional mechanism of antioxidation. The chemical structure and antioxidant activity of the oxide obtained by irradiating an ethanol solution of an equal mixture of butylated hydroxyanisole (BHA) and ethyl protocatechuate (EP) with the ultraviolet (UV) ray and its secondary decomposition were studied. The oxide was identified as 2,2′-dihydroxy-3-tert-butyl-5-methoxy-5′-carboethoxy-diphenyl ether which was a dimer of free radicals generated by BHA and EP. The resulting oxide showed a strong antioxidant activity on lard and methyl oleate in the stability testing by active oxygen method (AOM).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kurechi, T., Eisei Kagaku 13:191 (1967).
Kurechi, T., Ibid. 15:301 (1969).
Kurechi, T., and T. Taki, Yakugaku Zasshi 97:1174 (1977).
Kurechi, T., and U. Konno, oral presentation at 1976 Annual Meeting of the Pharmaceutical Society of Japan, Nagoya, April 1976.
D.H. Wheeler: Oil Soap (Chicago) 9:89 (1932).
Author information
Authors and Affiliations
About this article
Cite this article
Kurechi, T., Yamaguchi, T. Studies on the antioxidants: X. oxidation products of concomitantly used butylated hydroxyanisole and ethyl protocatechuate. J Am Oil Chem Soc 57, 216–219 (1980). https://doi.org/10.1007/BF02673943
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02673943