Abstract
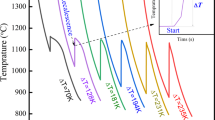
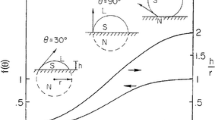
The ability of rapid conduction cooling to produce new non-equilibrium phases is examined with particular attention on solidification rate. By matching the rate of solidification calculated numerically from the heat flow balance with the crystal growth rate calculated by a kinetic equation, the kinetic undercooling for a given experimental condition is estimated. A limiting condition of cooling is presented in which the cooling rate is so large that the crystal growth cannot cover the whole sample body before it is cooled down to a low temperature where the growth rate of the crystal is negligibly small.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
ReferencesReferences
P. Duwez:Trans. ASM, 1967, vol. 60, pp. 607–33.
R. C. Ruhl:Mater. Sci. Eng., 1967, vol. 1, pp. 313–20.
R. W. Ruddle:The Solidification of Castings, 2nd ed., pp. 79–80, Institute of Metals, London, 1957.
H. S. Carslaw and J. C. Jaeger:Conduction of Heat in Solids, 2nd ed., pp. 282–96, Oxford Univ. Press, 1959.
G. E. Forsythe and W. R. Wasow:Finite-Difference Methods for Partial Differential Equations, pp. 88–139, J. Wiley and Sons Inc., New York, 1960.
J. W. Cahn,W. B. Hillig and G. W. Sears:Acta Met., 1964, vol. 12, pp. 1421–39.
J. Frenkel:Kinetic Theory of Liquids, p. 193, Dover Pub. Inc., New York, 1955.
M. E Fine:Phase Transformations in Condensed Systems, p. 49, Macmillan Co., New York, 1964.
J. R. Wilson:Metal. Rev., 1965, vol. 10, p. 552.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shingu, P.H., Ozaki, R. Solidification rate in rapid conduction cooling. Metall Trans A 6, 33–37 (1975). https://doi.org/10.1007/BF02673667
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02673667