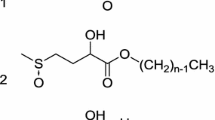
Abstract and Summary
Lactitol was oxyalkylated with propylene or ethylene oxide. The resulting ether-alcohols were transesterified with a series of fatty acid esters in order to produce long chain ether-esters of lactitol. These compounds were found to be more water soluble than related lactitol derivatives prepared by transesterification prior to oxyalkylation. Improved lime soap dispersant requirements (LSDR) and emulsification times were obtained from the new derivatives.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Scholnick, F., M.K. Sucharski, and W.M. Linfield, JAOCS 51:8 (1974).
Scholnick, F., G. Ben-et, M.K. Sucharski, E.W. Maurer, and W.M. Linfield, ibid. 52:7 (1975).
Borghetty, H.C., and C.A. Bergman, ibid. 27:88 (1950).
Osipow, L, F.D. Snell, W.C. York, and A. Finchler, Ind. Eng. Chem. 48:1459 (1956).
Author information
Authors and Affiliations
About this article
Cite this article
Scholnick, F., Linfield, W.M. Lactose- derived surfactants(III): Fatty esters of oxyalkylated lactito. J Am Oil Chem Soc 54, 430–432 (1977). https://doi.org/10.1007/BF02671026
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02671026