Abstract
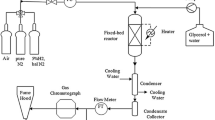
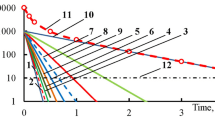
A simple approach to model the rate constants and selectivities for soybean oil hydrogenation with nickel catalyst is presented. The rate constant models were constructed on the basis of Arrhenius Law and Power series. The parameters were temperature, hydrogen pressure, concentration of catalyst and agitation expressed in terms of power per unit volume of oil. The models agree with the general knowledge in hydrogenation and the data were fitted fairly well by the models.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anderson, K. L. Petersson and N.H. Schoon, Acta Polytech. Scan., Chem. Incl. Met. Ser. 100:77 (1971).
Wisniak, J., and L.F. Albright, Ind. Eng. Chem. 53:375 (1961).
Hashimoto, K., M. Teramoto and S. Negata, J. Chem. Eng. Jpn. 4:150(1971).
Allen, R.R., JAOCS 55:751 (1978).
Glasstone, S., K.J. Laidler and H. Eyring, “The Theory of Rate Processes,” McGraw-Hill, New York, 1941, Ch. 1.
Johnstone, R.E., and M.W. Thring, “Pilot Plants, Models, and Scale-Up Methods in Chemical Engineering,” McGraw-Hill, New York, 1957, Ch. 8.
Allen, R.R., and J.E. Covey, Jr., JAOCS 47:494 (1970).
Smith, J.C., in “Perry’s Chemical Engineers’ Handbook,” 4th Edn, McGraw-Hill, New York, 1963, Ch. 19, p. 14.
Luong, H.T., and B. Volesky, AIChE J. 25:893 (1979).
Eldib, I.A., and L.F. Albright, Ind. Eng. Chem. 49:825 (1957).
Calderbank, P.H., in “Mixing: Theory and Practice, Vol. 2,” edited by V.W. Uhl and J.B. Gray, Academic Press, New York, 1967, p. 23.
Susu, A.A., A.F. Ogunye and C.O. Onyegbado, J. Appl. Chem. Biotechnol. 28:823 (1978).
Author information
Authors and Affiliations
Additional information
Presented at AOCS Annual Conference, New York, NY, April 1980.
About this article
Cite this article
Chen, A.H., Mclntire, D.D. & Allen, R.R. Modeling of reaction rate constants and selectivities in soybean oil hydrogenation. J Am Oil Chem Soc 58, 816–818 (1981). https://doi.org/10.1007/BF02665587
Issue Date:
DOI: https://doi.org/10.1007/BF02665587