Abstract
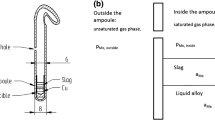
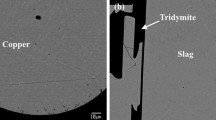
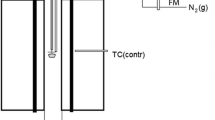
The thermodynamic behavior of bismuth in the chemical systems associated with copper processing is not well understood. This study was designed to further the understanding of the physical chemistry of bismuth in slags that have similar compositions to those found in copper extractive metallurgical processing. The silicate system investigated was the FeO-Fe2O3-SiO2 ternary system in which bismuth was dissolved using an isopiestic experimental technique. Bismuth vapor pressures of 1 • 10−5 atm and 7.5 • 10−4 atm were used, and the silicates were equilibrated with this vapor at temperatures of 1458 K and 1523 K. In these experiments, the slag composition was varied such thatP O 2 ranged from 10−12 to 10−8 atm. Bismuth was found to enter the silicate slag in both neutral and oxidic molecular forms. The oxidic form identified was that of BiO. The data suggest that the activity coefficient of neutral bismuth, γBi, is dependent on the solubility of that species in slag, even at the low concentrations observed in this study. It has been hypothesized, based on the large diameter of neutral Bi, that only a limited number of sites are available to accommodate neutral Bi, and that as the limit is approached γBi increases significantly. That hypothesis is shown to be consistent with the experimental results obtained in the present work as well as the results obtained by other investigators.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Nagamori, P. J. Mackey, and P. Tarassoff:Metall. Trans. B, 1975, vol. 6B, pp. 295–301.
J. B. See and W. J. Rankin: National Institute for Metallurgy, Report 2099, 1981.
E.A. Johnson, P.E. Sanker, L. L. Oden, and J.B. See: United States Bureau of Mines, RI 8655, 1982, 18 pp.
S. Goto, O. Ogawa, and I. Jimbo:Australia-Japan Extractive Metallurgy Symp. Proc, Australian Institute of Metallurgy, Sydney, Australia, 1980, pp. 127–32.
I. Jimbo, S. Goto, and O. Ogawa:Metall. Trans. B, 1984, vol. 15B, pp. 535–41.
A. Roine:Metall. Trans. B, 1987, vol. 18B, pp. 203–12.
A. Roine and H. Jalkanen:Metall. Trans. B, 1985, vol. 16B, pp. 129–41.
N. Asano and M. Wada:Suiyokaishi, 1968, vol. 16, pp. 385–88.
S. Arac and G. H. Geiger:Metall. Trans. B, 1981, vol. 12B, pp. 569–71.
T. Azakami and A. Yazawa:Can. Metall. Q., 1976, vol. 15, pp. 111–22.
V. S. Sibanda and E.H. Baker:IMM Trans. C, 1979, vol. 88, pp. C129–30.
V.J. Bode, J. Gerlach, and F. Pawlek:Erzmetal., 1971, vol. 24, pp. 480–85.
G.K. Sigworth and J.F. Elliott:Can. Metall. Q., 1974, vol. 13, pp. 455–61.
B. Predell and A. Emam:Zhurnal Metallic., 1973, vol. 64, pp. 496–501.
D.L. Hildebrand and K. H. Lau: Progress Report, Sept. 1979 to June 1980, NSF Grant DAR 79-12296, SRI Project Number 8933.
S. Arac: Ph.D. Thesis, University of Arizona, Tucson, AZ, 1980.
P. Taskinen:Scand. J. of Metal., 1982, vol. 11, pp. 150–54.
P. Taskinen and J. Niemela:Scand. J. of Metal., 1981, vol. 10, pp. 195–200.
D.C. Lynch and K.W. Schwanze:Can. Metall. Q., 1981,vol. 20, pp. 269–78.
R. F. Farrell, S. A. Matthes, and A. J. Mackie : United States Department of the Interior, Bureau of Mines, RI 8480, 1980, 14 pp.
L. N. Sidorov, I.I. Minayeva, E.Z. Zasorin, I.D. Sorokin, and A.Y. Borschchevskiy:High Temp. Sci., 1980, vol. 12, pp. 175–96.
V. S.Banand B.E. Knox:Chem. Phys., 1970, vol. 52, pp. 243–47.
V.K. Il’in:Zhur. Neorg. Khim., 1976, vol. 21, pp. 1645–48.
C.K. Kazenas, D.M. Chizhikov, Y. U. Tsvetkov, and M.V. Ol’shevskii:Doklady Akad. Nauk. SSSR, 1972, vol. 207, pp. 354–55.
O.M. Uy and J. Drowart:Trans. Faraday Soc, 1969, vol. 65, pp. 3221–30.
L.B. Pankratz: “Thermodynamic Properties of Elements and Oxides,” U.S. Bureau of Mines Bulletin 672, 1982.
F.J. Kohl, O.M. Uy, and K.D. Carlson:J. Chem. Phys., 1967, vol. 47, pp. 2607–76.
L. Rovner, A. Drowart, and J. Drowart:Trans. Faraday Soc., 1967, vol. 63, pp. 2906–11.
C. L. Sullivan, J. K. Prusaczyk, and K. D. Carlson:High Temp. Sci., 1972, vol. 4, pp. 212–22.
L.C. Wagner and R. T. Grimely:Chem. Phys. Letters, 1974, vol. 29, pp. 594–99.
G. H. Geiger and D. R. Poirier:Transport Phenomena in Metallurgy, Addison-Wesley, Reading, MA, 1973, p. 470.
G. D. Christian:Analytical Chemistry, 2nd ed., John Wiley & Sons, New York, NY, 1977, pp. 320–25.
A.E. Harvey, J.A. Smart, and E. S. Amis:Anal. Chem., 1955, vol. 27, pp. 26–29.
M. Nagamori and P.J. Mackey:Metall. Trans. B, 1977, vol. 8B, pp. 39–46.
L. Fang and D.C. Lynch:Metall. Trans. B, 1987, vol. 18B, pp. 181–87.
O. Kubaschewski and C.B. Alcock:Metallurgical Thermochemistry, 5th ed., Pergamon, Oxford, England, 1979, pp. 179–207.
R. B. Bird, W. E. Stewart, and E. N. Lightfoot:Transport Phenomena, John Wiley and Sons, New York, NY, 1960, pp. 567–72.
K. E. Grew and T. L. Ibbs:Thermal Diffusion in Gases, New York: Cambridge University Press, 1952.
H. Brown:Phys. Rev., 1940, vol. 58, pp. 661–62.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marschman, S.C., Lynch, D.C. Evaluation of the activity and molecular form of bi in cu smelting slags: Part I. ternary silicate slags. Metall Trans B 19, 627–641 (1988). https://doi.org/10.1007/BF02659155
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02659155