Abstract
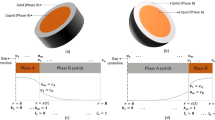
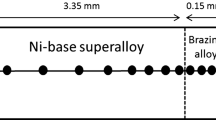
Detailed finite difference modeling of dissolution during transient liquid-phase (TLP) brazing of nickel using Ni-11 wt pct P and Ni-14 wt pct Cr-10 wt pct P filler metals is carried out assuminginfinite andfinite heating rates between the filler melting temperature (T m) and the brazing temperature (T b). Two conditions are modeled during brazing, namely, where the liquid width due to base metal dissolution isunrestrained (variable) and where the liquid width is keptconstant due to expulsion of excess liquid during the brazing process. The time required for completion of dissolution is proportional to the square of the filler metal thickness when an infinite heating rate betweenT m andT b is assumed. This occurs for both for constant and unrestrained liquid width situations. Also, the advance of the solid-liquid interface is approximately related to the reciprocal of the square root of the solute diffusivity in the liquid, except in the final stages of the dissolution process. When a multicomponent filler metal composition (Ni14 wt pct Cr-10 wt pct P) is used, the time for completion of dissolution exceeds that when using a simple binary filler metal composition. The phosphorous concentration in the liquid approaches that of the liquidus line when the heating rate betweenT m andT b decreases and thin filler metals are used. This tendency can be estimated using a homogenizing index (z) which combines the effects of filler metal thickness, diffusivity in the liquid, brazing temperature and heating rate, and a distribution index(a). When thick filler metals are used, lower heating rates produce more dissolution at temperatures betweenT m andT b and less dissolution at the final brazing temperature. Conversely, heating slowly when using thin filler metals produces less base metal dissolution (at any temperature) and negligible dissolution at the final brazing temperature. At very low heating rates, solidification can occur while heating fromT m toT b.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
D.S. Duvall, W.A. Owczarski, and D.F. Paulonis:Weld. J., 1974, vol. 53, pp. 203–14.
C. Nernst:Z. Phisikal Chem., 1904, vol. 47, pp. 52–55.
J.M. Lowell and B. Chalmers:Trans. TMS-AIME, 1959, vol. 215, pp. 499–506.
Y. Nakao, K. Nishimoto, K. Shinozaki, C.H. Kang, and Y. Hori:Q. J. Jpn. Weld. Soc., 1988, vol. 6, pp. 519–26.
I. Tuah-Poku, M. Dollar, and T.B. Massalski:Metall. Trans. A, 1988, vol. 19A, pp. 675–86.
M. Hansen:Constitution of Binary Equilibrium Diagrams, 2nd ed., McGraw-Hill, New York, NY, 1958.
R.P. Elliott:Constitution of Binary Alloys, 1st suppl. McGraw-Hill, New York, NY, 1965.
T.B. Massalski:Binary Alloy Phase Diagrams, ASM, Metals Park, OH, 1986.
H. Ikawa, Y. Nakao, and T. Isai:Trans. Jpn. Weld. Soc., 1979, vol. 1, pp. 24–29.
Y. Nakamura and H. Esaka:Tetsu-to-Hagané, 1981, vol. 67 (4), p. 140.
H.A. Majdic, D. Graf, and H. Schenek:Arch. Eisenhuettenwes., 1969, vol. 40, pp. 27–32.
P.L. Gruzin and V.V. Minal:Fiz. Met. Metalloved., 1963, vol. 16, pp. 551–54.
H. Nakagawa, C.H. Lee, and T.H. North: University of Toronto, Toronto, Canada, unpublished research, 1989.
Data Book of Metallurgy, Jpn. Inst. Met., Sendai, Japan, 1984, Maruzen.
T. Matsumiya, S. Mizoguchi, Y. Veshima, and H. Esaka:Trans. Iron Steel Inst. Jpn., 1984, vol. 24, pp. 873–80.
Author information
Authors and Affiliations
Additional information
Formerly Visiting Research Scientist, Department of Metallurgy and Materials Science, University of Toronto
Formerly Postdoctoral Fellow, Department of Metallurgy and Materials Science, University of Toronto
Rights and permissions
About this article
Cite this article
Nakagawa, H., Lee, C.H. & North, T.H. Modeling of base metal dissolution behavior during transient liquid-phase brazing. Metall Trans A 22, 543–555 (1991). https://doi.org/10.1007/BF02656822
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02656822