Abstract
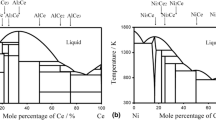
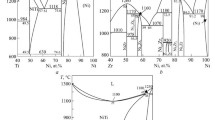
High-temperature (1425 to 2750 K) thermodynamic activities of one or both components of twenty-five binary alloys of a group IVB-VIB element (Ti, Zr, Hf, Nb, Ta, or W) with a platinum group element (Ru, Os, Ir, Pd, Pt, or Au) have been determined by equilibrating the alloy with the appropriate carbide and graphite, equilibrating with the nitride and nitrogen gas, or measuring the partial vapor pressure(s) thermogravimetrically or mass spectrometrically. The extraordinary stability of this class of transition metal alloy is attributed to a generalized Lewis-acid-base interaction involving valenced electrons, and the results of these investigations are interpreted within the context of this effect. Among the conclusions made are that a non-spherically-symmetrical crystal field significantly reduces the bonding effectiveness of certain valenced orbitals; the effect of the extent of derealization of these orbitals is also considered.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- A :
-
Area of Knudsen cell orifice
- a 1 :
-
Thermodynamic activity of speciesi
- dw/dt :
-
Mass loss per unit time
- E :
-
Relative ionization efficiency
- EPMA:
-
Electron probe microanalysis
- AG° f :
-
Standard free energy of formation
- ΔG i :
-
Partial molal free energy change for transferringi from its standard state (purei) to some mixture.
- Δ•G xs i :
-
Excess partial molal free energy change (= Δ~Gi +TΔ•S id i )
- ΔH° f :
-
Standard enthalpy of formation
- 1 + i :
-
Ion current for species i+
- k i :
-
Mass spectrometer pressure calibration constant for speciesi
- M:
-
Group IVB-VIB element
- M i :
-
Molecular weight of speciesi
- n :
-
Isotopic abundance
- P° :
-
Vapor pressure of speciesi over the pure condensed standard state
- PI:
-
Platinum group element
- R:
-
Ideal gas constant
- Δ•S id i :
-
Ideal (configurational) partial molal entropy change
- X i :
-
Mole fraction of speciesi
- XRD :
-
X-ray diffraction
- ±AX :
-
Uncertainty range corresponding to the reported value /A to the reported value⋅A
- γ i :
-
Electron multiplier gain or activity coefficient for speciesi, as indicated in text
- σ :
-
Ionization cross section
References
N. Engel:Ingenioeren, 1939, Sect. M, p. 101 and 1940, Sect. M, p. 1;Powder Met. Bull., 1954, vol. 7, pp. 8–18;ASM Trans., 1964, vol. 57, pp. 610–19;Acta Metall., 1967, vol. 15, pp. 557–63.
L. Brewer: inHigh Strength Materials, V. F. Zackay, ed., John Wiley and Sons, New York, NY, 1965, chap. 2, pp. 12–103.
L. Brewer:Acta Metall., 1967, vol. 15, pp. 553–56;Science, 1968, vol. 161, pp. 115–22.
V. Srikrishnan and P.J. Ficalora:Metall. Trans., 1974, vol. 5, pp. 1471–75.
L. Pauling:The Nature of the Chemical Bond, 3rd ed., Cornell Univ. Press, Ithaca, NY, 1960, pp. 93,410.
L. Brewer: inPhase Stability in Metals and Alloys, P. Rudman, J. Stringer, and R. I. Jaffe, eds., McGraw-Hill Book Co., New York, NY, 1967, pp. 39–61.
P.J. Meschter and W. L. Worrell:Metall. Trans. A, 1976, vol. 7A, pp. 299–305;Metall. Trans. A, 1977, vol. 8A, pp. 503–09.
L. Brewer and P. R. Wengert:Metall. Trans., 1973, vol. 4, pp. 83–104.
Ya. I. Gerassimov, V.I. Lavrentev, O. von Goldbeck, D.T. Livey, R. Ferro, and A. L. Dragoo:Tantalum: Phusico-Chemical Properties of its Compounds and Alloys, Special Issue No. 3, International Atomic Energy Agency, Vienna, 1972, pp. 44,49.
H. L. Schick:Thermodynamics of Certain Refractory Coupounds, Academic Press, New York, NY, 1966, vol. 2, pp. 83–84.
E. Storms, B. Calkin, and A. Yencha:High Temp. Science, 1969, vol. 1, pp. 430–55.
W.L. Worrell and J. Chipman:J. Phys. Chem., 1964, vol. 68, pp. 860–66.
E.K. Storms and J. Griffin:High Temp. Science, 1973, vol. 5, pp. 291–310.
P. J. Spencer, O. Kubaschewski-von Goldbeck, R. Ferro, R. Manazza, K. Girgis, and O. Kubaschewski:Hafnium: Physico-Chemical Properties of its Compounds and Alloys, Special Issue No. 8, International Atomic Energy Agency, Vienna, 1981, pp. 30–32.
JANAF Thermochemical Data, The Dow Chemical Co., Midland, MI: C ref. state, 1978; Hf ref. state, 1979; N2 ref. state, 1977.
V. I. Lavrentev, Ya. I. Gerassimov, P. Feschotte, D. T. Livey, O. von Goldbeck, H. Nowotny, K. Siefert, R. Ferro, and A. L. Dragoo:Niobium: Physico-Chemical Properties of its Compounds and Alloys, Special Issue No. 2, International Atomic Energy Agency, Vienna, 1968, p. 26.
R. Hultgren, P.D. Desai, D.T. Hawkins, M. Gleiser, and K. K. Kelley.Selected Values of the Thermodynamic Properties of Binary Alloys, ASM, Metals Park, OH, 1973, pp. 1170–76.
C.B. Alcock, K.T. Jacob, S. Zador, O. Kubaschewski-von Goldbeck, H. Nowotny, K. Siefert, and O. Kubaschewski:Zirconium: Physico-Chemical Properties of its Compounds and Alloys, Special Issue No. 6, International Atomic Energy Agency, Vienna, 1976, p. 28.
M. Battuelo and T. Ricolfi:High Temp.-High Prés., 1980, vol. 12, pp. 247–52.
Y. S. Touloukian and D. P. DeWitt:Thermophysical Properties of Matter, IFl/Plenum Data Corp., New York, NY, 1972, vol. 8, pp. 12,1058,1080,and 1086.
C.E. Holcomb: U.S.A.E.C. Oak Ridge Y-12 Plant, Report Y1887, 1973, reported in Ref. 32.
A. Amendola: Polytechnic Inst, of Brooklyn, NY, 1958, reported in Ref. 32.
T. F. Chase and E. F. Juenke: General Electric, Cincinnati, OH, 1968, reported in Ref. 32.
P.G. Cotter and J. A. Kohn:J. Am. Cer. Soc, 1954, vol. 37, pp. 415–20.
J. M. Bind and G. J. McCarthy: Pennsylvania State University, 1973, reported in Ref. 32.
E. Raub and G. Falkenburg:Metallwissenschaft und Technick, 1973, vol. 27, pp. 669–79.
A. E. Dwight and P. A. Beck:Trans. TMS-AIME, 1959, vol. 215, pp. 967–79.
J.W. Downey: Met. Div., Argonne Nat'l. Lab., 1964, reported in Ref. 32.
J.B. Darby, J.W. Downey, and L.J. Norton:TMS-AIME, 1963, vol. 227, pp. 1028–29.
A. Raman and K. Schubert:Z. Metalk., 1964, vol. 55, pp. 704–10.
J.W. Colby:MAGIC IV—α computer program for quantitative electron microprobe analysis, Bell Telephone Labs., Murray Hill, NJ, 1972.
JCPDS Powder Diffraction Files, International Centre for Diffraction Data, Swarthmore, PA, 1945 and after.
L. S. Birks:Electron Probe Microanalysis, 2nd ed., John Wiley and Sons, Inc., New York, NY, 1971, pp. 101–20.
J. I. Goldstein and J. W. Colby: inPractical Scanning Electron Microscopy, J.I. Goldstein and H. Yakowitz, eds., Plenum Press, New York, NY, 1975, pp. 435–89.
A. Taylor and N.J. Doyle:J. Less-Common Metals, 1967, vol. 13, pp. 413–30.
A. S. King:Trans. Am. Electrochem. Soc, 1929, vol. 56, pp. 97–110.
R. H. Hultgren, P. D. Desai, D. T. Hawkins, M. Gleiser, K. K. Kelley, and D. D. Wagman:Selected Values of Thermodynamic Properties of the Elements, ASM, Metals Park, OH, 1973.
K. S. Pitzer and L. Brewer: revision of G. N. Lewis and M. Randall,Thermodynamics, 2nd ed., McGraw-Hill Book Co., New York, NY, 1961, p. 246.
R. C. Paule and J. L. Margrave: inThe Characterization of High Temperature Vapors, J. L. Margrave, ed., John Wiley and Sons, Inc., New York, NY, 1967, pp. 130–51.
K. D. Carlson, P. W. Gilles, and R. J. Thorn:J. Chem. Phys., 1963, vol. 38, pp. 2725–35.
J. P. Hirth: inThe Characterization of High Temperature Vapors, J. L. Margrave, ed., John Wiley and Sons, Inc., New York, NY, 1967, pp. 453–72.
M. Knudsen:Ann. Physik, 1909, vol. 28, pp. 75–130.
G. L. Selman, P.J. Ellison, and A. S. Darling:Platinum Metals Review, 1970, vol. 14, pp. 14–20.
K. A. Gingerich: inCurrent Topics in Materials Science, E. Kaldis, ed., North Holland Publishing Company, New York, NY, 1980, vol. 6, pp. 345–462.
K. A. Gingerich:J. Chem. Phys., 1968, vol. 49, pp. 14–18.
K. Hilpert and K. A. Gingerich:Ber. Bunsenges Phys. Chem., 1980, vol. 84, pp. 739–45.
J.B. Mann:J. Chem. Phys., 1967, vol. 46, pp. 1646–51.
H. Kleykamp:J. Less-Common Metals, 1982, vol. 83, pp. 105–13.
Author information
Authors and Affiliations
Additional information
Formerly Graduate Student Research Assistant, Materials and Molecular Research Division, Lawrence Berkeley Laboratory, and Department of Chemistry, University of California, Berkeley
Rights and permissions
About this article
Cite this article
Gibson, J.K., Brewer, L. & Gingerich, K.A. Thermodynamics of several lewis-acid-base stabilized transition metal alloys. Metall Trans A 15, 2075–2085 (1984). https://doi.org/10.1007/BF02646841
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02646841