Abstract
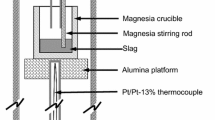
The kinetics of the evolution of SO2 gas from a liquid synthetic blast furnace -type slag (CaO-Al2O3-SiO2) in an atmosphere of O2 + Ar gas at a total pressure of 1 atm for 0.003 ≤ Po2 ≤ 1.00 atm has been studied in the range 1360 to 1460°C. The process has been followed by collecting and analyzing the SO2 as it forms, and also by observing the change in weight of the slag sample with time. The effect of slag composition has also been studied. For partial pressures of oxygen less than about 0.1 atm, the rate is very rapid and is controlled by transport in the gas phase. At greater values of Po2, the rate is much slower and is controlled by a chemical process. In the high Po2 region, the process is half-order with respect to the concentration of sulfur in the slag. This half-order dependence on sulfur concentration in the slag may be explained by an initial fast irreversible reaction to form two intermediate species which then decompose at equal rates to give the final products. Additions to the slag of iron or manganese oxides greatly accelerate the rate of evolution of SO2 at Po2 = 1.00 atm. This is interpreted to mean that a charge transfer process, possibly involving S2- and O2- ions, is rate-controlling at Po2 = 1 atm. It is also apparent that Fe2+ and Fe3+ (or Mn2+ and Mn3+) ions can act as charge carriers. Some measurements with actual industrial blast furnace slags are also reported.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
E. T. Turkdogan and M. L. Pearce:Trans. TMS-AIME, 1963, vol. 227, pp. 940- 49.
G. R. St. Pierre and J. Chipman:Trans. AIME, 1956, vol. 206, pp. 1474–83.
E. T. Turkdogan and L. S. Darken:Trans. TMS-AIME, 1961, vol. 221, pp. 464- 74.
J. F. Elliott, M. Gleiser, and V. Ramakrishna:Thermochemistry for Steelmaking, Vol. II, p. 707, Addison-Wesley, Inc., Reading, Massachusetts, 1963.
K. J. Laidler:Chemical Kinetics, 2nd ed., pp. 262–95, McGraw-Hill Book Co., Inc., New York, New York, 1965.
Author information
Authors and Affiliations
Additional information
Formerly Visiting Scientist, Massachusetts Institute of Technology
Rights and permissions
About this article
Cite this article
Pelton, A.D., See, J.B. & Elliott, J.F. Kinetics of evolution of SO2 from hot metallurgical slags. Metall Trans 5, 1163–1171 (1974). https://doi.org/10.1007/BF02644328
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02644328