Abstract
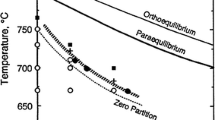
The dissolution of Fe3C in ferrite has been shown to be controlled by a slow first order interfacial reaction. The rate constant,K, is Arrhenius in behavior and involves two activation enthalpies; one applies to a jump process with a short relaxation time, another to a long relaxation time process which is tentatively identified as a change in kink density on ledges in the interface. The heat of solution of Fe3C in ferrite also appears in the exponential. A single process, activated jump theory is not adequate to explain the behavior ofK with temperature; a dissolution model based on a ledge-double kink mechanism appears to be in agreement with experiment. These features point to significant differences between the mechanism of interphase boundary migration and grain boundary migration. In addition, the Fe3C-ferrite interface is felt to be at equilibrium with respect to carbon but not iron, leading to small departures from stoichiometry in the Fe3C at the interface.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
F. V. Nolfi, Jr., P. G. Shewmon, and J.S. Foster:Trans. TMS-AIME, 1969, vol. 245, pp. 1727–33.
F. V. Nolfi, Jr., P. G. Shewmon, and J. S. Foster:Met. Trans., 1970, vol. 1, pp. 789–800.
H. Margenau and G. M. Murphy:The Mathematics of Physics and Chemistry, D. Van Nostrand Company, Inc., 1956.
L. S. Darken and R. W. Gurry:Physical Chemistry of Metals, McGraw Hill Book Co., New York, 1953.
J.C. Swartz:Trans. TMS-AIME, 1969, vol. 245, pp. 1083–92.
J. C. Swartz:Trans. TMS-AIME, 1967, vol. 239, pp. 68–75.
K. Löcke and H. P. Stüwe:Recovery and Recrystallization of Metals, pp. 171–210, Interscience Publishers, 1963.
R. P. Smith:J. Am. Chem. Soc., 1948, vol. 70, pp. 2724–29.
R. W. Heckel and R. L. DeGregorio:Trans. TMS-AIME, 1965, vol. 233, pp. 2001–11.
W. B. Pearson:Handbook of Lattice Spacings and Structures of Metals-Vol. 2, pp. 427–28, Pergamon Press, 1967.
L.J.E.Hofer: U.S. Bur. Mines Bull. 631,1966.
L. S. Darken and R. M. Fisher:Decomposition of Austenite by Diffusional Processes, pp. 249–94, Interscience Publishers, 1962.
P. M. Kelly and J. Nutting:J. Iron Steel Inst., 1961,vol. 197,pp. 199–211.
H. Gleiter:Acta Met., 1969, vol. 17, pp. 565–73.
J. P. Hirth:Energetics in Metallurgical Phenomena-Volume II, pp. 1–52, Gordon and Breach Science Publishers, 1965.
W. K. Burton, N. Cabrera, and F. C. Frank:Phil. Trans. Roy. Soc, 1951, vol. 243A, pp. 299–358.
K.J. AustandJ.W. Rutter:Trans. TMS-AIME, 1959,vol. 215, pp. 119–27.
K. J. Aust and J. W. Rutter:Trans. TMS-AIME, 1959, vol. 215, pp. 820–31.
B.B. Rath and Hsun Hu:Trans. TMS-AIME, 1969,vol. 245,pp. 1577–85.
D. Tumbull:AIME Trans., 1951, vol. 191, pp. 661–65.
H. Gleiter:Acta Met., 1969, vol. 17,pp. 853–62.
Author information
Authors and Affiliations
Additional information
Formerly Graduate Student, Department of Metallurgy and Materials Science, Carnegie-Mellon University, Pittsburgh, Pa.
This paper is based on a thesis submitted by FRANK V. NOLFI, JR. in partial fulfillment of the requirements of the degree of Doctor of Philosophy at Carnegie-Mellon University.
Rights and permissions
About this article
Cite this article
Nolfi, F.V., Shewmon, P.G. & Foster, J.S. The dissolution kinetics of Fe3C in ferrite — A theory of interface migration. Metall Trans 1, 2291–2298 (1970). https://doi.org/10.1007/BF02643447
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02643447