Abstract
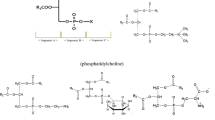
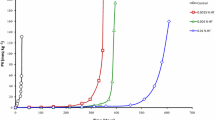
The antioxidant properties of phospholipids (PL) in a refined salmon oil model system were measured by determining changes in the 2-thiobarbituric acid number and decreases in the ratio of docosahexaenoic acid (DHA)/palmitic acid (22:6/16:0) of a fish oil system incubated at 180°C for up to 3 h. The more phosphatidylcholine (PC) added to the oil system, the higher the oxidative stability obtained. The order of effectiveness of commercial phospholipids in inhibiting oxidation and the loss of polyunsaturated fatty acids was as follows: sphingomyelin (SPH)=lysophosphatidylcholine (LPC)=phosphatidylcholine (PC)=phosphatidylethanolamine (PE)>phosphatidylserine (PS)>phosphatidylinositol (PI)>phosphatidylglycerol (PG)>control salmon oil. Nitrogen containing PL, including PE, PC, LPC and SPH, were equally effective in exerting greater antioxidant properties than PS, PG and PI. The inverse relationship observed between the oxidation index (C22:6/C16:0) and color intensity for treatments following 2 h of heating suggests that Maillard-type reaction products may have contributed to the oxidative stability of PL-supplemented fish oils.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lea, C.H.,J. Sci. Fd. Agric. 8:1 (1957).
Nasner, A., inLipid Oxidation-Biological and Food Chemical Aspects: Contributions at a Lipid Forum/Sik Symposium, edited by R. Marcuse, Scandinavian Forum for Lipid Research and Technology, Gotenborg, Sweden, 1986, p. 187.
List, G.R., and J.P. Friedrich,J. Am. Oil Chem. Soc. 66:98 (1989).
Hudson, B.J.F., and S.E.O. Mahgoub,J. Sci. Fd. Agric. 32:208 (1984).
Yamaguchi, K., and M. Toyomizu,Bull. Soc. Sci. Fish. 50:1897 (1984).
Kashima, M., G.S. Cha, Y. Isoda, J. Hirano and T. Miyazawa,J. Am. Oil Chem. Soc. 68:119 (1991).
Privett, O.S., and F.W. Quackenbush, Ibid.:225 (1954).
Jewell, N.E., and W.W. Nawar, Ibid.:398 (1980).
Husain, S.R., J. Terao and S. Matsushita, inAmino-Carbonyl Reactions in Food and Biological Systems, edited by M. Fugimaki, M. Namiki and H. Kato, Elsevier Press, New York, NY, 1984, p. 301.
Porter, W.L., inRecent Trends in Food Applications of Antioxidants, edited by M.G. Simic, and M. Karel, Plenum Press, New York, NY, 1980, p. 295.
Moberger, L., inLipids Oxidation-Biological and Food Chemical Aspects: Contributions at a Lipid Forum/Sik Symposium, Gotenborg, Sweden, edited by R. Marcuse, Scandinavian Forum for Lipid Research and Technology, Gotenborg, Sweden, 1986, p. 114.
King, M.F., and L.C. Boyd, Abstract, Institute of Food Technologists’ Annual Conference, Anaheim, CA, 1990, p. 219.
Widicus, W.A., and J.R. Kirk,Assoc. Off. Anal. Chem. 62:637 (1979).
Kaduce, T., K.C. Norton and A.A. Spector,J. Lipid Res. 24:1398 (1983).
Morrison, W.R., and L.M. Smith, Ibid.:600 (1964).
Sampugna, J., L.A. Pallansch, M.E. Enig and M. Keeney,J. Chromatogr. 24:245 (1982).
Ke, P.J., and A.W. Woyewoda,Analytica Chimica Acta 106:279 (1979).
Shono, T., and M. Toyomizu,Bull. Jap. Soc. Sci. Fish 22:290 (1972).
Helwig, J.T., and K.A. Council,SAS User’s Guide, edited by SAS Institute, Inc., Cary, NC, 1982.
SAS,Statistical Analysis System. User’s Guide: Statistics, SAS Institute Inc., Cary, NC, 1988.
Tsai, L., and L.M. Smith,Lipids 6:196 (1972).
Hildebrand, D.H., J. Terao and M. Kito,J. Am. Oil Chem. Soc. 61:552 (1984).
Corlis, G.A., and J.R. Dugan,Lipids 5:846 (1970).
Bratkowska, I.,Acta Alim. Pol. 4:256 (1978).
Evans, C.D., P.M. Cooney, C.R. Scholfield and H.J. Dutton,J. Am. Oil Chem. Soc. 31:295 (1954).
Author information
Authors and Affiliations
About this article
Cite this article
King, M.F., Boyd, L.C. & Sheldon, B.W. Antioxidant properties of individual phospholipids in a salmon oil model system. J Am Oil Chem Soc 69, 545–551 (1992). https://doi.org/10.1007/BF02636106
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02636106