Summary
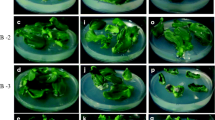
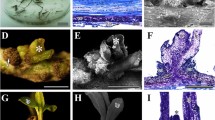
A simple and rapid procedure for direct organogenesis from root nodulelike structures of alfalfa (Medicago sativa L.) line SGg, spontaneously induced on growth regulator-free Gamborg (B5) medium, was developed. Prolific adventitious shoot initiation was obtained using a combination of 1.0 mg/liter TIBA and 0.5 mg/liter 2iP. Transfer of shoots to a medium containing 0.5 mg/liter ABA and reduced concentration of TIBA (0.5 mg/liter) before rooting markedly stimulated shoot development. Regenerated shoots rooted easily and revealed the early appearance of nodulelike structures on basal medium (B5) lacking growth regulators. Analysis of endogenous growth regulator levels of SGg roots maintained on growth regulators free media, showed that spontaneous shoot appearances was correlated with high cytokinin-to-auxin ratios.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ainley, W. M.; McNeil, K. J.; Hill, J. W., et al. Regulatable endogenous production of cytokinins up to “toxic” levels in transgenic plants and plant tissues. Plant Mol. Biol. 22:13–23; 1993.
Ammirato, P. V. Hormonal control of somatic embryo development from cultured cells of caraway. Plant Physiol. 59:579–586; 1977.
Atanassov, A. I.; Brown, C. D. Plant regeneration from suspension cultures and mesophyll protoplasts ofMedicago sativa L. Plant Cell Tissue Organ Cult. 3:149–152; 1984.
Bingham, E.; Hurley, L.; Kaatz, D., et al. Breeding of alfalfa which regenerates from callus tissue culture. Crop. Sci. 15:719–729; 1975.
Binns, A. N.; Labriola, J.; Black, R. C. Initiation of auxin autonomy inNicotiana glutinosa cells by the cytokinin-biosynthesis gene fromA. tumefaciens. Planta 171:539–548; 1987.
Brown, C. D.; Atanassov, A. I. Role of genetic background in somatic embryogenesis inMedicago. Plant Cell Tissue Organ Cult. 4:111–112; 1985.
Christianson, M.; Warnick, D. Organogenesis in vitro as a developmental process. XXII Int. Hort. Cong. 83rd ASHS Annual Meeting; Davis, CA; 1986.
Crouch, M.; Sussex, M. Development of storage protein synthesis inBrassica napus L. embryosin vitro andin vivo. Planta 153:64–74; 1981.
De Bruijn, F. M.; Szabados, I.; Schell, J. Chimeric genes and transgenic plants are used to study the regulation of genes involved in plant-microbe interactions (Nodulin genes). Dev. Genet. 11:182–196; 1990.
Denchev, P.; Velcheva, M.; Atanassov, A. A new approach to direct somatic embryogenesis inMedicago. Plant Cell Rep. 10:338–342; 1991.
Dijak, M.; Simmonds, D. Micritubule organization during early direct embryogenesis from mesophyll protoplasts ofMedicago sativa L. Plant Sci. 58:183–191; 1988.
Gamborg, O.; Miller, R.; Ojima, H. Nutrient requirements of suspension cultures of soybean roots cells. Exp. Cell. Res. 50:151–158; 1968.
Hirsch, A. M.; Bhuvaneswarri, R. V.; Torrey, J. G., et al. Early nodulin genes are induced in alfalfa root outgrowths elicited by auxin transport inhibitors. Proc. Natl. Acad. Sci. USA 86:1244–1248; 1989.
Ivanova, A.; Eshkenazy, M.; Van Onckelen, H. Development of immunoassays for endogenous cytokinin and indole-3-acetic-acid determination. Biotechnology & Biotech. Equipment 2:7–12; 1994.
Joshi, P. A.; Gaetanoanolles, G.; Graham, E. T., et al. Ultrastructure of transfer cells in spontaneous nodules of alfalfa (Medicago sativa) Protoplasma 172:2–4, 64–76; 1993.
Long, S.Rhizobium-legume symbiosis: life together in the underground. Cell 56:203–214; 1989.
Long, S.; Cooper, J. Overview of symbiosis. In: Palacios R.; Verma, A. P. S., eds. Molecular genetics of plant-microbe interactions. Minnesota: APS Press, St. Paul, MN: 1988:163–178.
McCoy, T. J.; Bingham, E. T. Regeneration of diploid alfalfa plants from cell grown in suspension culture. Plant Sci. Lett. 10:59–66; 1977.
Mitten, D.; Sato, S.; Skokun, T. In vitro regenerative potential of alfalfa germplasm sources. Crop. Sci. 24:943–945; 1984.
Nolan, R. E.; Rose, R. J.; Gorst, J. R. Regeneration ofMedicago truncatula from tissue culture: increased somatic embryogenesis using explants from regenerated plants. Plant Cell Rep. 8:278–281; 1989.
Prinsen, B.; Rudelshein, P.; Van Onckelen, H. Extraction, purification and analysis of endogenous indole-3-acetic acid and abscisic acid. In: Negrutiu, I.; Chazt-Chhetzi, G., eds. A Laboratory Guide for Cellular and Molecular Plant Biology 1991:192–209.
Rolfe, B.; Greesshoff, P. Genetic analysis of legume nodule initiation. Annu. Rev. Plant Physiol. Plant Mol. Biol. 39:297–319; 1988.
Shepard, J. Abscisic acid-enhanced shoot initiation in protoplastderived calli of potato. Plant Sci. Lett. 18:327–333; 1980.
Smigocki, A.; Owens, L. Cytokinin-to-auxin ratios and morphology of shoots and tissues transformed by a chimeric isopentenyl transferase gene. Plant Physiol. 91:808–811; 1989.
Song, J.; Sorensen, E.; Ziang, G. Direct embryogenesis from single mesophyll protoplasts in alfalfa (Medicago sativa L.) Plant Cell Rep. 9:21–25; 1990.
Truchet, G.; Barker, D.; Camut, S., et al. Alfalfa nodulation in the absence ofRhizobium. Mol. Gen. Genet. 219:65–68; 1989.
Van De Wiel, C.; Norris, J. H.; Bochenek, B., et al. The early nodulin transcript Enod 2 is located in the nodule parenchyma (inner cortex) of pea and soybean root nodules. Plant Cell 2:1009–1017; 1990.
Vlahova, M. G.; Atanassov, A. I. Establishment of the system for regeneration from different genotypes of alfalfa cultivated in vitro. In the 2nd Youth Gen. Conf. BAS (Sofia) 1984:18–23.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sarul, P., Vlahova, M., Ivanova, A. et al. Direct shoot formation in spontaneously occurring root pseudonodules of alfalfa (Medicago sativa L.). In Vitro Cell Dev Biol - Plant 31, 21–25 (1995). https://doi.org/10.1007/BF02632221
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02632221