Abstract
Tripalmitin, 1- and 2-lauryl dipalmitin and 1- and 2-oleyl dipalmitin were subjected to thermal oxidation at 200C in the presence of air for various lengths of time. The triglycerides showed a loss in weight, and an increase in carbonyl hydroxyl and acid values. The I.V. increased in the case of saturated triglycerides and decreased in the case of unsaturated triglycerides.
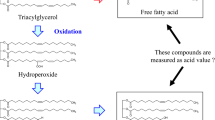
Hydrolysis of the ester linkage between glycerol and fatty acid was found to occur during thermal oxidation of the type and position of the fatty acid in the triglyceride molecule. The fatty acids released from the triglyceride by hydrolysis were found either to be oxidized further to short chain fatty acids, or were oxygenated with the introduction of a carbonyl or hydroxyl group in the molecule. Moreover, the unsaturated fatty acid in the triglyceride molecule was found to be oxidized more readily than the saturated fatty acid.
A hydroxy fatty acid with a carbon number of 13.5 on a diethylene glycol succinate column was isolated from oxidized tripalmitin and was also found to occur in the free fatty acid fraction of oxidized tripalmitin, 1-lauryl, 2–3 dipalmitin, and 1-oleyl, 2–3 dipalmitin. The presence of laurie or oleic acid in the 2-position of the triglyceride prevented the formation of this acid, which suggested that it is an oxidation product of palmitic acid.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Holman, R. T., “Progress in the Chemistry of Fats and Other Lipids,” 2, 51, Academic Press Inc., New York (1954).
Powers, P. O., Ind. Eng. Chem.41, 304 (1949).
Bateman, L., Quart. Revs., Chem. Soc. (London)8, 147 (1954).
Farmer, E. H., and Sutton, D. A., J. Chem. Soc. 122 (1943).
Atherton, D., and Hilditch, T. P., J. Chem. Soc., 204 (1943).
Ross, J., Gebhart, A. I., and Gerecht, J. F., J. Amer. Chem. Soc.,71, 282 (1949).
Swift, C. E., Dollear, F. G., and O'Conner, R. T., Oil and Soap,23, 355 (1946).
Skellon, J. H., J. Chem. Soc., 2020 (1950).
Zuidema, H. H., Chem. Rev.,38, 197 (1946).
Shoruigin, P. P., and Kreshkov, A. P., J. Gen Chem. (USSR),3, 825 (1933); C.A. 28, 6106 (1934).
Johnson, O. C., and Kummerow, F. A., J. Am. Oil Chemists' Soc.,34, 407 (1957).
Ellis, Rex, Gaddis, A. M., and Currie, G. T., Anal. Chem.,30, 475 (1958).
Perkins, E. G., Food Tech.,14, 508 (1960).
Hartman, L., J. Chem. Soc., 3572 (1957).
Ast, H. J., and Vander Wal, R. J., J. Am. Oil Chemists' Soc.,38, 67 (1961).
Hornstein, I., Alford, J. A., Elliott, L. E., and Crowe, P. F., Anal. Chem.,32, 540 (1960).
Bush, M. T., and Densen, P. M., Anal. Chem.,20, 121 (1948).
Official and Tentative Methods of the American Oil Chemists' Society, Editor: V. C. Mehlenbacher, Chicago, Illinois, 1946.
Smith, W. T., Jr., and Shriner, R. L., The Examination of New Organic Compounds, J. Wiley and Sons, Inc., New York (1956).
Lappin, G. R., and Clark, L. C., Anal. Chem.,23, 541 (1951).
Bryant, W. M. D., and Smith, D. M., J. Amer. Chem. Soc.,57, 57 (1935).
Lundberg, W. O., and Chipault, J. R., J. Amer. Chem. Soc.,69, 833 (1947).
Mirva, T. K., Mikolojezak, K. L., Earle, F. R., and Wolff, I. A., Anal. Chem.,32, 1739 (1960).
Woodford, F. P., and Van Gent, C. M., J. Lipid Research,1, 188 (1960).
Nobori, H., J. Soc. Chem. Ind. (Japan), 45, Suppl. Binding, 453 (1942).
Ramanathan, V., Sakuragi, T., and Kummerow, F. A., J. Am. Oil Chemists' Soc.,36, 244 (1959).
Ellis, G., J. Soc. Chem. Ind.,45, 193 (1926).
Moreno, J. M., and Lopez, J. M. Comp. rend. 27th Congr. Intrn. Chem. Ind. Brussels, 1954, 3, Industrie Chem. Belge, 20, Spec. no. 717 (1955). (C.A. 50:9037i.)
Skellon, J. H., and Taylor, C. G., J. Chem. Soc., 1433 (1953).
Zeinalof, B. K., Trudy Inst. Khim. Akad. Nauk, Azerbaidzhan, S.S.R.,13, 91 (1954).
Author information
Authors and Affiliations
Additional information
Portion of a thesis presented by J. G. Endres as partial fulfillment of the requirements for the degree of Ph.D. in food technology.
Funds for partial support of these studies were made available by the National Institute of Health, Grant A-1671.
About this article
Cite this article
Endres, J.G., Bhalerao, V.R. & Kummerow, F.A. Thermal oxidation of synthetic triglycerides I. composition of oxidized triglycerides. J Am Oil Chem Soc 39, 118–121 (1962). https://doi.org/10.1007/BF02631685
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02631685