Summary
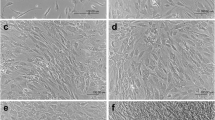
Cultures of human bone cells were established, maintained, and characterized with respect to several metabolic parameters. These studies were undertaken with a view to using the bone culture system as a means of studying mechanisms of bone metabolism. The donor patients' ages ranged from 1 to 90 years and their disease states included congenital limb anomalies, exostosis, and osteo- and rheumatoid arthritis. Cultures were maintained up to 5 months. The osteoblast-like character of these cells was confirmed with the use of measurements applied to bone cells from other systems. Analyses showed that (a) the cells' appearance resembled that of cultured osteoblasts from other animal sources, b) intracellular cAMP was stimulated by human parathyroid hormone, c) osteocalcin was detected in the medium of all tested bone cell cultures and its production was found to be stimulated by 1,25-dihydroxycholecalciferol, and d) newly synthesized collagen was almost exclusively type I.
In contrast, cultures of human fibrolasts, established in one instance from tissue specimens of the same donor patient, grew faster, reached a higher limiting density, and produced a greater proportion of type III collagen than the corresponding bone cells. Furthermore, fibroblasts did not accumulate osteocalcin in their culture medium.
The conditions described in this report to maintain human bone cells in culture should provide a suitable test system to study the regulation of human bone metabolism.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Peck WA, Binge SJ, Fedak SA (1964) Bone cells: biochemical and biological studies after enzymatic isolation. Science 146:1476–1477
Peck WA, Carpenter J, Messinger K, DeBra D (1973) Cyclic 3′5′ adenosine monophosphate in isolated bone cells: response to low concentrations of parathyroid hormone. Endocrinology 92:692–697
Hefley TJ, Stern PH (1982) Isolation of osteoclasts from fetal rat long bones. Calcif Tissue Int 34:480–487
Wong GL, Cohn DV (1974) Separation of parathyroid hormone and calcitonin sensitive cells from non-responsive bone cells. Nature 252:713–715
Osdoby P, Martini HC, Caplan AI (1982) Isolated osteoclasts and their presumed progenitor cells, the monocyte, in culture. J Exp Zool 224:331–344
Majeska RJ, Rodan SB, Rodan GA (1980) Parathyroid hormone responsive clonal cell lines from rat osteosarcoma. Endocrinology 107:1494–1498
Hohmann EL, Tashjian AH (1984) Functional receptors for vasoactive intestinal peptide on human osteo-sarcoma cells. Endocrinology 114:1321–1327
Mills BG, Singer FR, Weiner LP, Holst PA (1979) Long-term culture of cells from bone affected by Paget's disease. Calcif Tissue Int 29:79–87
Wergedaal JE, Baylink DJ (1982) Isolation and culture of human bone cells. Calcif Tissue Int Suppl 34:S16 (abstract)
Beresford JN, MacDonald BR, Gowen M, Couch M, Akad D, Sharpe PT, Gallagher JA, Poser JW, Russell RGG (1983) Further characterization of a system for the culture of human bone cells. Calcif Tissue Int 35:637 (abstract)
Gowen M, Beresford J, Gallagher J, Wood DD, Russell RGG (1983) Actions of interleukin I on bone metabolism in vitro. Calcif Tissue Int 35:642 (abstract)
Wergedaal JE, Farley JR, Baylink DJ (1983) Direct effect of fluoride on proliferation and alkaline phosphatase of human bone cells. Calcif Tissue Int 35:652 (abstract)
Cohn DV, Wong GL (1979) Isolated bone cells. In: Simmons DJ, Kunin AS (eds) Skeletal research. Academic Press, New York, London, pp 3–20
Gundberg CM, Lian JB, Gallop PM (1983) Measurements of gammacarboxyglutamate and circulating osteocalcin in normal children and adults. Clin Chim Acta 128:1–8
Goldberg RL, Parrott DP, Kaplan SR, Fuller GC (1980) Effect of gold sodium thiomalate on proliferation of rheumatoid synovial cells and on collagen synthesis is tissue culture. Biochem Pharmacol 29:869–876
Neville DM (1971) Molecular weight determination of protein dodecyl-sulfate complex by gel electrophoresis in a continuous buffer system. J Biol Chem 246:6328–6334
Owen M (1980) The origin of bone cells in the postnatal organism. Arthritis Rheum 23:1073–1080
Schwartz ER (1979) Cartilage cell and organ culture. In: Simmons DJ, Kunin AS (eds) Skeletal research. Academic Press, New York, London, pp. 61–80
Rose GG (1970) Atlas of vertebrate cells in tissue culture. Academic Press, New York
Binderman I, Duksin S, Harell A, Katzir E, Sachs L (1974) Formation of bone tissue in culture from isolated bone cells. J Cell Biol 61:427–439
Goldring SR, Dayer JM, Russell RG, Mankin HJ, Krane SM (1978) Response to hormones of cells cultured from giant cell tumors of bone. J Clin Endocrinol Metab 46:425–433
Austin LA, Heath LA (1981) Calcitonin. N Engl J Med 304:269–278
Chambers TJ, Magnus CJ (1982) Calcitonin alters behaviour of isolated osteoclasts. J Pathol 136:27–40
Guenther HL, Felix R, Fleisch H (1984) Comparative study of deflazacort, a new synthetic corticosteroid, and dexamethasone on the synthesis of collagen in different rat bone cell populations and rabbit articular chondrocytes. Calcif Tissue Int 36:145–152
Nishimoto SK, Price PA (1980) Secretion of the vitamin K-dependent protein of bone by rat osteosarcoma cells. J Biol Chem 255:6579–6583
Price PA, Lothringer JW, Baukol SA, Reddi AH (1981) Developmental appearance of the vitamin K-dependent protein of bone during calcification. J Biol Chem 256:3781–3784
Weinstein RS, Gundberg CM (1984) Serum osteocalcin levels reflect bone formation in patients with metabolic bone disease. J Clin Invest (in press)
Price PA, Baukol SA (1980) 1,25-dihydroxyvitamin D3 increases synthesis of the vitamin K-dependent bone protein by osteosarcoma cells. J Biol Chem 255:11660–11663
Gundberg CM, Cole DE, Lian JB, Reade TM, Gallop PM (1983) Serum osteocalcin in the treatment of inherited rickets with 1,25-dihydroxyvitamin D3. J Clin Endocrinol Metab 56:1063–1067
Ecarot-Charrier B, Glorieux FH (1982) Isolation of osteoblast-like cells from mouse calvaria. Calcif Tissue Int Suppl 34:S5 (abstract)
Whitson SW, Harrison WR, Knox JN, Unhold GE, Robey PG, Fisher LW, Termine JD (1983) Bovine bone cells synthesize osteonectin and other bone-specific marker proteins in vitro. Calcif Tissue Int 35:653 (abstract)
Miller EJ (1973) A review of biochemical studies on the genetically distinct collagens of the skeletal system. Clin Orthop Res 92:260–280
Cheung HS, Singer FR, Mills B, Nimni ME (1980) In vitro synthesis of normal bone (type I) collagen by bone of Paget's disease patients. Proc Soc Exp Biol Med 163:547–552
Rowe DW, Kream BE (1982) Regulation of collagen synthesis in fetal rat calvaria by 1,25-dihydroxyvitamin D3. J Biol Chem 257:8009–8015
Scott DM, Kent GN, Cohn DV (1980) Collagen synthesis in cultured osteoblast-like cells. Arch Biochem Biophys 201:384–391
Uitto J, Bauer EA, Eisen AZ (1979) Increased biosynthesis of triple helical type I and type III procollagens associated with unaltered expression of collagenase by skin fibroblasts in culture. J Clin Invest 64:921–930
Fraser J, Lancaster GA, Sorvi CR (1983) Secreted collagen ratios in normal human and osteogenesis imperfecta skin fibroblasts. Conn Tiss Res 11:57–67
Shaprio JR, Triche T, Rowe CW, Munabi A, Cattell HS, Schlesinger S (1983) Osteogenesis imperfecta and Paget's disease of bone. Biochemical and morphological studies. Arch Intern Med 143:2250–2257
Prockop DJ, Kivirikko KI, Tuderman L, Guzman NA (1979) The biosynthesis of collagen and its disorders (two parts). N Engl J Med 301:13–23, 77–85
Steinmann BU, Reddi AH (1980) Changes in synthesis in types I and III collagen during matrix-induced endochondral bone formation in rat. Biochem J 186:919–924
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Auf'mkolk, B., Hauschka, P.V. & Schwartz, E.R. Characterization of human bone cells in culture. Calcif Tissue Int 37, 228–235 (1985). https://doi.org/10.1007/BF02554868
Issue Date:
DOI: https://doi.org/10.1007/BF02554868