Abstract
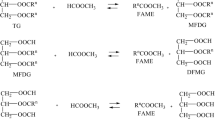
Methyl fatty esters derived from vegetable oils are a promising fuel for direct injection diesel engines. This study’s purpose was to identify a heterogeneous catalyst to selectively produce methyl fatty esters from low erucic rapeseed oil. Most experiments were at atmospheric pressure and approximately the corresponding boiling point temperature of the mixture, 60–63 C. However, the catalytic activity of an anion exchange resin was tested at 200 C and 68 atm (1000 psig) and at 91 C and 9.2 atm (135 psig). All samples were analyzed by thin layer chromatography with samples from the elevated temperature and pressure experiments also analyzed by mass spectroscopy. The most promising catalyst examined was CaO·MgO. The activities of the catalysts CaO and ZnO appear to be enhanced with the addition of MgO, therefore the transesterification reaction mechanism may be, in this instance, bifunctional. The anion exchange resin catalyst at 200 C and 68 atm generated substantial amounts of both methyl fatty esters and straight-chain hydrocarbons, even though these reactions did not go to completion. At 91 C and 9.2 atm the cracking also occurred but at a substantially reduced rate, and no transesterification was noted.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pryde, E.H., Veg. Oil Fuels, Proc. Int. Conf., Fargo, ND, 1982, pp. 101–105.
Wagner, L.E., “Methyl Ethyl and Butyl Soybean Oil Esters-Alternate Fuels for Compression Ignition Engines,” Master’s thesis, Kansas State Univ., Manhattan, KS, 1983.
Pishinger, G.H.; A.M. Falcon; R.W. Siekmann, and F.R. Fernandes, Veg. Oil Fuels, Proc. Int. Conf., Fargo, ND, 1982, pp. 198–208.
Hassett, D.J., Veg. Oil Diesel Fuel Semin. III, Peoria, IL, 1983, pp. 72–77.
Hawkins, C.S., and J. Fuls, Veg. Oil Fuels, Proc. Int. Conf., Fargo, ND, 1982, pp. 184–197.
Ventura, L.M.; A.C. Nascimento and W. Bandel, Ibid., pp. 394–400.
Freedman, B., and E.H. Pryde, Veg. Oil Fuels, Proc. Int. Conf., Fargo, ND, 1982, pp. 117–122.
Kusy, P.F., Ibid., pp. 127–137.
Fuls, J., and F.J.C. Hugo, Third Int. Conf. Energy Use Manage., Berlin, 1981, Pergamon Press, 1981, pp. 1595–1602.
Nye, M.J.; T.W. Williamson; S. Deshpande; J.H. Schrader; W.H. Snively; T.P. Yurkewich, and C.L. French, JAOCS 60:1598 (1983).
Freedman, B.; E.H. Pryde, and T.L. Mounts, JAOCS 60:737 (1983).
Groggins, P.H., “Processes in Organic Synthesis,” 3rd ed., McGraw-Hill, New York, 1947, pp. 624–626.
Sridharan, R., and I.M. Mathai, J. Sci. Ind. Res. 33:178 (1974).
Tanabe, K., “Solid Acids and Bases, Their Catalytic Properties,” Academic Press, New York, 1970, pp. 2, 5–7, 35–37, 54–55, 107.
Rindt, J.R., “Heterogeneous Catalysis of Solvent Refined Lignite to Obtain Chemical Feed Stocks,” Master’s thesis, Univ. of North Dakota, Grand Forks, ND, 1979, pp. 52–53.
Wu, W.R.K., and H.H. Storch, “Hydrogenation of Coal and Tar,” Bureau of Mines, Washington, DC, 1968, p. 63.
Dykstra, G.J., and S.C. Sorenson, Veg. Oil Diesel Fuel Semin. III, Peoria, IL, 1983, pp. 38–44.
Author information
Authors and Affiliations
About this article
Cite this article
Peterson, G.R., Scarrah, W.P. Rapeseed oil transesterification by heterogeneous catalysis. J Am Oil Chem Soc 61, 1593–1597 (1984). https://doi.org/10.1007/BF02541639
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02541639