Abstract
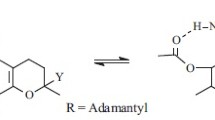
There is evidence that an addition compound of oxidizeddl-alpha-tocopherol and linoleic acid is formed when the components are absorbed in mixed monolayer on silica gel at a molecular ratio of 1∶20, and subjected to heating in air at 80C. A relatively nonpolar tocopheryl quinone is also formed in smaller amounts. These are the major tocopherol oxidation products isolated in this system and do not correspond to any known to the authors. The addition compound has about the same mobility as linoleic acid in most thin layer chromatography (TLC) and chromatographic systems, but can be isolated by successive chromatography on silica and gel filtration on Sephadex LH-20. It yields a single spot in TLC in several systems. The elemental analysis is reproducible and consistent with a simple addition compound of linoleic acid and bivalently oxidized tocopherol. The compound has a carboxyl group which can be esterified. The ester has about the same TLC mobility as methyl linoleate. The molecular weight of the ester is 722.6. The UV spectrum shows a single peak, 2 ETOHmax =3000 Å, E=4.74. The IR spectrum shows a very strong chroman ether band at 9.12 μ, a strong methyl band at 7.24 μ and carboxyl but no hydroxyl absorption. The NMR spectrum shows, in contrast to that of tocopherol, a reduction in aromatic methyl protons, a carboxyl proteon exchangeable with deuterium oxide, but no hydroxy proton. The compound does not reduce Emmerie-Engel reagent prior to treatment with concentrated hydriodic acid, nor do the ether-extractable products after such treatment. The present data are consistent with an addition product whose bridging group is a new chroman ring.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
AOCS Meeting, Chicago, October 1967, Abstract No. 89; also W.L. Porter, L.A. Levasseur, A.S. Henick and J.I. Jeffers, Lipids, in press.
AOCS Meeting, New York, October 1968, Abstract No. 117; also W. L. Porter, A.S. Henick, J.I. Jeffers and L.A. Levasseur, Lipids, in press.
Nelan, D.R., and C.D. Robeson, J. Amer. Chem. Soc. 84:2963–2965 (1962).
Skinner, W.A., and P. Alaupovic, Science 140:803–805 (1963).
Csallany, A.S., and H.H. Draper, Arch. Biochem. Biophys. 100:335–337 (1963).
Csallany, A.S., M. Chiu and H.H. Draper, Lipids 5:63–70 (1970).
Strauch, B.D., H.M. Fales, R.C. Pittman and J. Avigan, J. Nutr. 97:194–202 (1969).
Schudel, von P., H. Mayer, J. Metzger R. Ruegg and O. Isler, Helv. Chim. Acta 46:636–649 (1963).
Dürckheimer, W., and L.A. Cohen, Biochem. Biophys. Res. Commun. 9:262–265 (1962).
Martius, C., and H. Eilingsfeld, Justius Liebigs Annalen 607:159–168 (1957).
Goodhue, C.T., and H.A. Risley, Biochem. Biophys. Res. Commun. 17:549–553 (1964).
Nilsson, J. Lars, G., J.O. Branstad, H. Sievertsson, Acta. Pharm. Suecica 5:509–516 (1968).
Skinner, W.A., and R.M. Parkhurst, J. Org. Chem. 31:1248–1251 (1966).
Stahl, E., “Thin Layer Chromatography,” Academic Press, New York, 1965, p. 475.
Parker, W.E. and W.D. McFarlane, Can. J. Res. 18B:405–409 (1940).
Bishov, S.J., and A.S. Henick, JAOCS 43:477 (1966).
Quisenberry, K.S., T.T. Scolman and A.O. Nier, Phys. Rev. 102:1071–1075 (1956).
Metcalfe, L.D., and A.A. Schmitz, Anal. Chem. 33:363–364 (1961).
Shriner, R.L., R.C. Fuson and D.Y. Curtin, “The Systematic Identification of Organic Compounds,” Fourth Edition, John Wiley & Sons, Inc. New York, 1959, p. 116.
Green, J., S. Marcinkiewicz and D. McHale, J. Chem. Soc. C. Org. 1966 (6), 1422–1427.
Simon, E.J., A. Eisengart, L. Sundheim and A.T. Milhorat, J. Biol. Chem. 221:807–817 (1956).
Bovey, F.A., “Nuclear Magnetic Resonance Spectroscopy,” Academic Press, New York, 1969, p. 249.
Author information
Authors and Affiliations
About this article
Cite this article
Porter, W.L., Levasseur, L.A. & Henick, A.S. An addition compound of oxidized tocopherol and linoleic acid. Lipids 6, 1–8 (1971). https://doi.org/10.1007/BF02536368
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02536368