Abstract
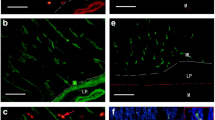
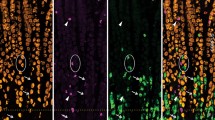
Immunohistochemical localization of GABAB-receptors was demonstrated in the rat gastrointestinal tract using a monoclonal antibody (GB-1) raised against the purified GABAB-receptor. Immunoreactive staining for GABAB-receptors was found in some populations of endocrine, muscular and neuronal components in the stomach and gut wall. Positive mucosal epithelial, probably endocrine, cells were distributed throughout the stomach and intestine. Double immunostaining indicated that such positive cells for GABAB-receptors often co-possessed serotonin in the small intestine but not in the gastric body. In the muscular layer of the digestive canal, positive staining was seen as dotty granules punctuated on the surface of muscle fibers. In the enteric nervous system, positive neuronal somata were found in both submucosal and myenteric ganglia throught the entire canal extending from the stomach to the rectum. This is the first report to visualize the cellular localization of GABAB-receptors in the gastrointestinal system of the rat, and should provide a fundamental basis for future studies on gastrointestinal functions regulated by GABAB-receptors.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ong, J., and Kerr, D. I. B. 1990. GABA-receptors in peripheral tissues. Life Sci. 46:1489–1501.
Saito, N., and Tanaka, C. 1986. Immunohistochemical demonstration of GABA containing neurons in the guinea pig ileum using purified GABA antiserum. Brain Res. 376:78–84.
Hills, J. M., Jessen, K. R., and Mirsky, R. 1987. An immunohistochemical study of the distribution of enteric GABA-containing neurons in the rat and guinea-pig intestine. Neuroscience 22:301–312.
Furness, J. B., Trussell, D. C., Pompolo, S., Bornstein, J. C., Maley, B. E., and Storm-Mathisen, J. 1989. Shapes and projections of neurons with immunoreactivity for gamma-aminobutyric acid in the guinea-pig small intestine. Cell Tissue Res. 256:293–301.
Gilon, P., Campistron, G., Geffard, M., and Remacle, C. 1988. Immunohistochemical localization of GABA in endocrine cells of the rat entero-pancreatic system. Biol. Cell 62:265–273.
Davanger, S., Ottersen, O. P., and Storm-Mathisen, J. 1989). GABA-immunoreactive cells in the rat gastrointestinal epithelium. Anat. Embryol. 179:221–226.
Bowery, N. G., Doble, A., Hill, D. R., Hudson, A. L., Shaw, J. S., Turnbull, M. J., and Warrington, R. 1981. Bicuculline-insensitive GABA receptors on peripheral autonomic nerve terminals. Eur. J. Pharmacol. 71:53–70.
Kuriyama, K., Hirouchi, M., and Nakayasu, H. 1993. Structure and function of cerebral GABAA and GABAB receptors. Neurosci. Res. 17:91–99.
Ong, J., and Kerr, D. I. B. 1983. GABAA-and GABAB-receptormediated modification of intestinal motility. Eur. J. Pharmacol. 86: 9–17.
Giotti, A., Spagnesi, S., and Zilletti, L. 1983. GABAA and GABAB receptor-mediated effects in guinea-pig ileum. Br. J. Pharmacol. 78:469–478.
Krantis, A., and Harding, R. K. 1987. GABA-related actions in isolated in vitro preparations of the rat small intestine. Eur. J. Pharmacol. 141:291–298.
Del Tacca, M., Blandizzi, C., Bernardini, M. C., Martelli, F., and Della-Longa, A. 1989. GABA-A mediated gastrin release induced by baclofen in the isolated vascularly perfused rat stomach. Experientia 45:734–736.
Gentilini, G., Franchi-Micheli, S., Pantalone, D., Cortesini, C., and Zilletti, L. 1992. GABAB receptor-mediated mechanisms in human intestine in vitro. Eur. J. Pharmacol. 217:9–14.
Erdö, S. L., De Vincentis, G., and Amenta, F. 1990. Autoradiographic localization of [3H] muscimol binding sites in rat stomach: evidence for mucosal GABAA receptors. Eur. J. Pharmacol. 175:351–354.
Nakayasu, H., Mizutani, H., Hanai, K., Kimura, H., and Kuriyama, K. 1992. Monoclonal antibody to GABA binding protein, a possible GABAB receptor. Biochem. Biophys. Res. Commun. 182:722–726.
Nakayasu, H., Nishikawa, M., Mizutani, H., Kimura, H., and Kuriyama, K. 1993. Immunoaffinity purification and characterization of γ-aminobutyric acid (GABA)B receptor from bovine cerebral cortex. J. Biol. Chem. 268:8658–8664.
Takeuchi, Y., Kimura, H., and Sano, Y. 1982. Immunohistochemical demonstration of the distribution of serotonin neurons in the brainstem of the rat and cat. Cell Tiss. Res. 224:247–267.
Aimi, Y., Fujimura, M., Vincent, S. R., and Kimura, H. 1991. Localization of NADPH-diaphorase-containing neurons in sensory ganglia of the rat. J. Comp. Neurol. 306:382–392.
Jessen, K. R., Hills, J. M., and Limbrick, A. R. 1988. GABA immunoreactivity and3H-GABA uptake in mucosal epithelial cells of the rat stomach. Gut 29:1549–1556.
Schwörer, H., Racké, K., and Kilbinger, H. 1989. GABA receptors are involved in the moduration of the release of 5-hydroxytryptamine from the vascularly perfused small intestine of the guineapig. Eur. J. Pharmacol. 165:29–37.
Krantis, A., Kerr, D. I. B., and Dennis, B. J. 1986. Autoradiographic study of the distribution of [3H]γ-aminobutyrate-accumulating neural elements in guinea-pig intestine: evidence for a transmitter function of g-amnobutyrate. Neuroscience 17:1243–1255.
Jessen, K. R., Hills, J. M., Dennison, M. E., and Mirsky, R. 1983. γ-aminobutyrate as an autonomic neurotransmitter: release and uptake of [3H]γ-aminobutyrate in guinea pig large intestine and cultured enteric neurons using physiological methods and electron microscopic autoradiography. Neuroscience 10:1427–1442.
Bult, H., Boeckxstaens, G. E., Pelckmans, P. A., Jordaens, F. H., Van Maercke, Y. M., and Herman, A. G. 1990. Nitric oxide as an inhibitory non-adrenergic non-cholinergic neurotransmitter. Nature 345:346–347.
Hope, B. T., Michael, G. J., Knigge, K. M., and Vincent, S. R. 1991. Neuronal NADPH diaphorase is a nitric oxide synthase. Proc. Natl. Acad. Sci. 88:2811–2814.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue dedicated to Dr. Kinya Kuriyama.
Rights and permissions
About this article
Cite this article
Nakajima, K., Tooyama, I., Kuriyama, K. et al. Immunohistochemical demonstration of GABAB receptors in the rat gastrointestinal tract. Neurochem Res 21, 211–215 (1996). https://doi.org/10.1007/BF02529137
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02529137