Abstract
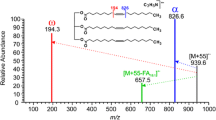
Conditions for the mass spectrometric analysis of triacylglycerols,via direct exposure probe, with ammonia negative-ion chemical ionization were optimized. Triacylglycerols were most favorably ionized, using the reactant gas pressure of approximately 8500 mtorr at the ion source temperature of 200°C with the instrumentation used. Abundant [M-H]− ions were produced under these conditions without the formation of [M+35]− cluster ions, which would interfere with the molecular weight region of triacylglycerols in the spectra. A rapid desorption of the sample from the probe wire is recommended, using a relatively high heating rate (approximately 40 mA s−1), to minimize thermal degradation of unsaturated molecules and the reducing effect of double bonds on the mass spectrometric response of triacylglycerols. Furthermore, the abundance of [M-H]− ion was enhanced by rapid heating, which we found to be important to improve the sensitivity. The appropriate amount of sample applied to the rhenium wire was in the region of 50–300 ng for one determination, i.e., only a few nanograms of a single triacylglycerol is required for production of a reliable spectrum. The reproducibility of the method was good as demonstrated with standards and a raspberry seed oil sample. The described mass spectrometric method is a fast and potentially useful tool for semiquantitative determination of triacylglycerol mixtures of various fats and oils. The discrimination, caused by differences in molecular size and unsaturation of triacylglycerols with 50 to 56 acyl carbons, was negligible under our optimized ionization conditions, thus, no correction factors were needed. These findings have not yet been verified with instruments in other laboratories. However, the present study shows how the analysis of triacylglycerols can be improved, regardless of the instrument, by optimization of the analytical conditions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CI:
-
chemical ionization
- EI:
-
electron ionization
- GC:
-
gas chromatography
- HPLC:
-
high-performance liquid chromatography
- MS:
-
mass spectrometry
- TAG:
-
triacylglycerols
References
Ryhage, R., and Stenhagen, E. (1960) Mass Spectrometry in Lipid Research,J. Lipid Res. 1, 361–390.
Barber, M., Merren, T.O., and Kelly, W. (1964) The Mass Spectrometry of Large Molecules I. The Triglycerides of Straight Chain Fatty Acids,Tetrahedron Letts 1063–1067.
Hites, R.A. (1970) Quantitative Analysis of Triglyceride Mixtures by Mass Spectrometry,Anal. Chem. 42, 1736–1740.
Hites, R.A. (1975) Mass Spectrometry of Triglycerides,Methods Enzymol. 35, 348–359.
Lin, Y.Y., and Smith, L.L. (1984) Chemical Ionization Mass Spectrometry of Steroids and Other Lipids,Mass Spectrom. Rev. 3, 319–355.
Murphy, R.C. (1993)Mass Spectrometry of Lipids, pp. 189–211, Plenum Press, New York.
Murata, T. (1977) Analysis of Triglycerides by Gas Chromatography/Chemical Ionization Mass Spectrometry,Anal. Chem. 49, 2209–2213.
Murata, T., and Takahashi, S. (1977) Qualitative and Quantitative Chemical Ionization Mass Spectrometry of Triglycerides,Anal. Chem. 49, 728–731.
Marai, L., Myher, J.J., and Kuksis, A. (1983) Analysis of Triacylglycerols by Reversed-Phase High-Performance Liquid Chromatography with Direct Liquid Inlet Mass Spectrometry,Can. J. Biochem. Cell Biol. 61, 840–849.
Kuksis, A., Marai, L., and Myher, J.J. (1983) Strategy of Glycerolipid Separation and Quantitation by Complementary Analytical Techniques,J. Chromatogr. 273, 43–66.
Kuksis, A., Marai, L., and Myher, J.J. (1991) Reversed-Phase Liquid Chromatography-Mass Spectrometry of Complex Mixtures of Natural Triacylglycerols with Chloride-Attachment Negative Chemical Ionization,J. Chromatogr. 588, 73–87.
Kuksis, A., Marai, L., and Myher, J.J. (1991) Plasma Lipid Profiling by Liquid Chromatography with Chloride-Attachment Mass Spectrometry,Lipids 26, 240–246.
Kallio, H., and Currie, G. (1993) Analysis of Low Erucic Acid Turnip Rapeseed Oil (Brassica campestris) by Negative-Ion Chemical Ionization Tandem Mass Spectrometry. A Method Giving Information on the Fatty Acid Composition in Positionssn-2 andsn-1,3 of Triacylglycerols,Lipids 28, 207–215.
Kallio, H., and Currie, G. (1993) Analysis of Natural Fats and Oils by Ammonia Negative-Ion Tandem Mass Spectrometry—Triacylglycerols and Positional Distribution of Their Acyl Groups, in.CRC Handbook of Chromatography, Analysis of Lipids (Mukherjee, K.D., Weber, N., and Sherma, J., eds.) pp. 435–458, CRC Press, Boca Raton.
Harrison, A.G. (1983)Chemical Ionization Mass Spectrometry, CRC Press, Boca Raton.
Westmore, J.B., and Alauddin, M.M. (1986) Ammonia Chemical Ionization Mass Spectrometry,Mass Spectrom. Rev. 5, 381–465.
Ways, P., and Hanahan, D.J. (1964) Characterization and Quantitation of Red Cell Lipids in Normal Man,J. Lipid Res. 5, 318–328.
Schulte, E., Höhn, M., and Rapp, U. (1981) Mass Spectrometric Determination of Triglyceride Patterns of Fats by Direct Chemical Ionization Technique (DCI),Fresenius Z. Anal. Chem. 307, 115–119.
Author information
Authors and Affiliations
About this article
Cite this article
Laakso, P., Kallio, H. Optimization of the mass spectrometric analysis of triacylglycerols using negative-ion chemical ionization with ammonia. Lipids 31, 33–42 (1996). https://doi.org/10.1007/BF02522407
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02522407