Abstract
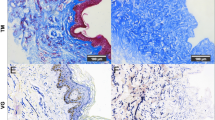
Higher-density inoculation of fibroblasts into a three-dimensional scaffold should accelerate wound healing after skin implantation. This study attempted to develop tissue-engineered skin with a higher density of fibroblasts. We first attempted to fabricate three-dimensional high-cell-density aggregates (spheroids) of normal human fibroblasts for application to tissue-engineered skin. Our method consisted of rotational shaking with nontreated dishes, decreasing fibroblast-meterial interactions, and augmenting cell-cell interaction. To prompt aggregate formation, the medium was supplemented with insulin, dexamethasone, ascorbic acid, and basic fibroblast growth factors that potentiate secretion of extracellular matrices. Under such improved conditions, fibroblasts were able to form spheroidal aggregates within 24 to 36h of rotational culture. Although the formed aggregates were irregular in shape and were composed of only several cells after 12h, they became almost spheroidal after 24h. The aggregates grew even more round after 36h, and their surfaces became smooth. After 36h of rotational culture, the fibroblast aggregates were collected and reinoculated onto a biodegradable mesh composed of polyglycolic acid coated with collagen. The aggregates were trapped in the material and became attached after 24h. Finally, because transforming growth factor-β3 (TGF-β3) is known to accelerate wound healing, we conducted a semiquantitative analysis of TGF-β3 mRNA in both the fibroblast monolayers (two-dimensional culture) and the aggregates (three-dimensional culture). Analysis of TGF-β3 mRNA expression showed that mRNA expression was greater in the fibroblasts of aggregates than in a monolayer. Therefore, our newly developed dermal graft is expected to accelerate wound healing faster than conventional grafts.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cooper ML, Hansbrough JF, Spielvogel RL, Cohen R, Bartel RL, Naughton G. In vivo optimization of a living dermal substitute employing cultured human fibroblasts on a biodegradable polyglycolic acid or polyglactin mesh. Biomaterials 1991;12:243–248
Hansbrough JF, Cooper ML, Cohen R, Spielvogel R, Greenleaf G, Bartel RL, Naughton G. Evaluation of a biodegradable matrix containing cultured human fibroblasts as a dermal replacement beneath meshed skin grafts on athymic mice. Surgery 1992;111:438–446
Mansbridge J, Liu K, Patch R, Symons K, Pinney E. Three-dimensional fibroblast culture implant for the treatment of diabetic foot ulcers: metabolic activity and therapeutic range. Tissue Eng 1998;4:403–414
Naughton GK, Bartel R, Mansbridge J. Tissue engineering in the epithelial system. In: Atala A, Mooney D (eds) Synthetic biodegradable polymer scaffolds. New York: Birkhäuser, 1997;121–148
Pollak R, Edington H, Jensen JL, Kroeker RO, Gentzkow GD, The Dermagraft Diabetic Ulcer Study Group. A human dermal replacement for the treatment of diabetic foot ulcers. Wounds 1997;9:175–183
Kawai K, Suzuki S, Tabata Y, Ikada Y, Nishimura Y. Accelerated tissue regeneration through incorporation of basic fibroblast growth factor-impregnate gelatin microspheres into artificial dermis. Biomaterials 2000;21:489–499
Sakai Y, Furukawa K, Suzuki M. Immobilization and long-term albumin secretion of hepatocyte spheroids rapidly formed by rotational tissue culture methods. Biotechnol Techniques 1992;6:527–532
Sakai Y, Naruse K, Nagashima I, Muto T, Suzuki M. Large-scale preparation and functions of porcine hepatocytes spheroids. Int J Artif Organs 1996;19:294–301
Sakai Y, Naruse K, Nagashima I, Muto T, Suzuki M. A new bioartificial liver using porcine hepatocyte spheroids in high-cell-density suspension perfusion culture: in vitro performance in synthesized culture medium and 100% human plasma. Cell Transplant 1999;8:531–541
Appling WD, O'Brien WR, Johnston DA, Duvic M. Synergistic enhancement of type I and III collagen production in cultured fibroblasts by transforming growth factor-beta and ascorbate. FEBS Lett 1989;250:541–544
Gehrke TA, Walboomers XF, Jansen JA. Influence of transforming growth factor-beta, on fibrous capsule formation around microgrooved subcutaneous implants in vivo. Tissue Eng 2000;6:505–517
Dai W, Saltzman WM. Fibroblast aggregation by suspension with conjugates of poly (ethylene glycol) and RGD. Biotechnol Bioeng 1996;50:359–356
Takezawa T, Yamazaki M, Mori Y, Yoshizato K. Morphological and immuno-cytochemical characterization of a hetero-spheroid composed of fibroblasts and hepatocytes. J Cell Sci 1992;101:495–501
Takezawa T, Mori Y, Yonaha T, Yoshizato K. Characterization of morphology and cellular metabolism during the spheroid formation by fibroblasts. Exp Cell Res 1993;208:430–441
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Furukawa, K.S., Ushida, T., Sakai, Y. et al. Tissue-engineered skin using aggregates of normal human skin fibroblasts and biodegradable material. J Artif Organs 4, 353–356 (2001). https://doi.org/10.1007/BF02480031
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02480031