Abstract
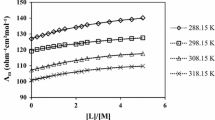
Compounds of the series LixLa(2−x)/3TiO3 (x=0.14, 0.23, 0.32, 0.35) as well as materials which are substituted with Pr, Tb, Cr and Fe were investigated by the coulometric titration method. The equilibrium potential as a function of δLi as determined by the coulometric titration measurements was fitted using the random solution model which takes into account the ionic configurational entropy and the electronic contributions to the chemical potential of lithium.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
6. References
L.L. Kochergina, N.B. Khakhin, N.V. Porotnikov and K.I. Petrov, Zhurnal Neorganicheskoi Khimii,29, 879 (1984).
A.G. Belous, G.N. Novitskaya, S.V. Polyanetskaya and Yu.I. Gornikov, Izv. Akad. Nauk SSSR, Neorg. Mater.,23, 470 (1987).
Y. Inaguma, L. Chen, M. Itoh, T. Nakamura, T. Uchida, H. Ikuta and M. Wakihara, Solid State commun.,86, 689 (1993).
Y. Inaguma, L. Chen, M. Itoh and T. Nakamura Solid State Ionics,70/71, 196 (1994).
H. Kawai and J. Kuwano, J. Electrochem. Soc.141, L78 (1994).
A.D. Robertson, S. Garcia Martin, A. Coats and A.R. West, J. Mater. Chem.5, 1405–1412 (1995).
I.D. Raistrick, A.J. Mark and R.A. Huggins, Solid State Ionics,5, 351–354 (1981).
M. Klingler, W.F. Chu and W. Weppner, Paper presented at the 11th International Conference on Solid State Ionics, November 16–21, 1997, Honolulu, Hawaii, USA.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Klingler, M., Chu, W.F. & Weppner, W. Coulometric titration of substituted LixLa(2−x)/3 TiO3 . Ionics 3, 289–291 (1997). https://doi.org/10.1007/BF02375631
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02375631