Abstract
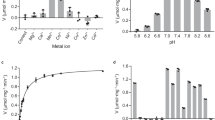
In order to evaluate the functional differences that may exist in human lactate dehydrogenase (LDH) isoenzymes widely used for clinical examination the kinetic and thermodynamic properties of the lactate to pyruvate reaction that they catalize were examined. Small but significant differences in the kinetic properties of the three isoenzymes were observed. The difference in the rate constants might affect the activity measurement of the individual isoenzyme as the initial velocity for the L-P reaction catalyzed will not be the same for an equal amount of enzyme. Equilibrium constants for the overall reaction in the presence and absence of pyruvate have been determined. On the basis of transition-state theory, the standard enthalpy and free-energy changes for formation of ternary activated complex were positive, while the standard entropy change was negative. Although the standard free-energy change was the same for activation by the three isoenzymes, the enthalpy and entropy changes for the LDH-3-catalyzed reaction were different from the respective values for others. A large positive value for the free-energy change and a negative value for the entropy change indicated unfavorable production of the activated complex (K sup╪infeq. =1.89×10-16). The enzyme appears to stabilize and retain the activated complex until it dissociates into the products.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
AlbertyR. A. (1953).J. Am. Chem. Soc. 75, 1928–1932.
AndersonS. R., FloriniJ. R., and VestlingC. S. (1964).J. Biol. Chem. 239, 2991–2997.
BradfordM. M. (1976).Anal. Biochem. 72, 248–254.
CohenL., DjordjevichJ., and JacobsenS. (1966).Med. Clin. North. Am. 50, 193–209.
DavisB. J. (1964).Ann. N.Y. Acad. Sci. 121, 404–427.
FondyT. P., PesceA., FreedbergI., StolzbachF. E., and KaplanN. O. (1964).Biochemistry 3, 522–530.
GutfreundH. (1971). inProbes of Structure and Function of Macromolecules and Membranes (B.Chance, T.Yonetani, and A. S.Mildvan, eds.), Vol. 2. Academic Press, Orlando, Florida, pp. 119–131.
HakalaM. T., GlaidA. J., and SchwertG. W. (1956).J. Biol. Chem. 221, 191–209.
HammesY. (1932) inEnzyme Catalysis and Regulation. Academic Press, Orlando, Florida, pp. 48–50.
HolbrookJ. J., and GutfreundH. (1973).Fed. Biochem. Soc. Lett. 31, 157–169.
HolbrookJ. J., LiljasA., SteindelS. J., and RossmanM. (1975). inThe Enzymes (BoyerP. E., ed.), Vol. XI, part A. Academic Press, Orlando, Florida, pp. 191–292.
HowellB. F., McCumeS., and SchafferR. (1979).Clin. Chem. 25, 269–272.
HunterI., AttockB., and PalmerT. (1983).Clin. Chim. Acta 135, 73–78.
MarketC. L., and MollerF. (1959).Proc. Natl. Acad. Sci. U.S.A. 6, 753–763.
MenonM. P., NambiarG. K., and NairR. M. G. (1985).J. Radidoanal. Nucl. Chem. 92, 123–132.
MenonM. P., MillerS., and TaylorB. S. (1986).J. Chromatogr. Biomed. Appl. 378, 450–455.
NairR. M. G., MenonM. P., and SagelJ. (1985).Clin. Physiol. Biochem. 3, 157–165.
PageM. I. (1980).Int. J. Biochem. 11, 331–335.
PlaceA. R., and PowersD. A. (1984).J. Biol. Chem. 259, 1309–1318.
ValkirsA. (1978).Comp. Biochem. Physiol. 59A, 31–36.
VasudevanG., MercerD. W., and VaratM. A. (1978).Circulation 57, 1055–1057.
WinerA. D., and SchwertG. M. (1958).J. Biol. Chem. 231, 1065–1083.
WackerW. E. C., UlmerD. D., and VallieB. L. (1956).N. Engl. J. Med. 255, 449–456.
ZeweV., and FrommH. J. (1962).J. Biol. Chem. 237, 1668–1675.
ZeweV., and FrommH. J. (1965).Biochemistry 4, 782–792.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Menon, M.P., Hunter, F.R. & Miller, S. Kinetic studies on human lactate dehydrogenase isoenzyme-catalyzed lactate-to-pyruvate reaction. J Protein Chem 6, 413–429 (1987). https://doi.org/10.1007/BF02343339
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02343339