Abstract
Purpose: The kinetics of tumor regression during administration of chemotherapy has relevance to the timing of surgery. The aim of this study was characterization of the time course of primary tumor regression in initially unresectable rhabdomyosarcoma, hepatoblastoma, and neuroblastoma patients. We also estimated the total cell number in the primary tumor at diagnosis.
Methods: Tumor volumes of 24 pediatric patients with either unresectable rhabdomyosarcoma, hepatoblastoma, or neuroblastoma were determined by using computerized three-dimensional reconstruction from serial computed tomography (CT) scans during chemotherapy. Cell densities were calculated by counting cell numbers in high-power fields and dividing by area and section thickness. Cell number at diagnosis was then calculated.
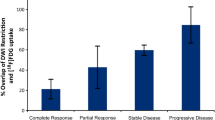
Results: Median tumor volumes at diagnosis were 175 cc, 748 cc, and 738 cc for rhabdomyosarcoma, neuroblastoma, and hepatoblastoma, respectively. The median tumor cell counts were 31, 68, and 59 × 1010 cells/tumor for rhabdomyosarcoma, neuroblastoma, and hepatoblastoma, respectively. The tumor regression was most rapid during the first two cycles, and little change in volume was observed after three cycles.
Conclusion: Rapid initial reduction in primary tumor volume with chemotherapy was observed in rhabdomyosarcoma, neuroblastoma, and hepatoblastoma. These data suggest that second-look resection may be feasible after two to three cycles of chemotherapy. This hypothesis may be tested by randomizing the timing of second-look surgical intervention.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wheatley JM, Rosenfeld NS, Heller G, Feldstein D, La Quaglia MP. Validation of a technique of computer-aided tumor volume determination.J Surg Res 1995;59:621–6.
Maurer HM, Gehan EA, Beltangady M, et al. The Intergroup Rhabdomyosarcoma Study-II.Cancer 1993;71:1904–22.
Matsumura M, Atkinson JB, Hays DM, et al. An evaluation of the role of surgery in metastatic neuroblastoma.J Pediatr Surg 1988;23:448–53.
La Quaglia MP, Kushner BH, Heller G, Bonilla MA, Lindsley KL, Cheung NK. Stage IV neuroblastoma diagnosed at more than 1 year of age: gross total resection and clinical outcome.J Pediatr Surg 1994;29:1162–6.
Sitarz A, Finklestein J, Grosfeld J, et al. An evaluation of the role of surgery in disseminated neuroblastoma: a report from the Children's Cancer Study Group.J Pediatr Surg 1983;18:147–51.
Friedman MA, Resser KJ, Marcus FS. How accurate are computed tomographic scans in assessment of changes in tumor size?Am J Med 1983;75:193–8.
Cohen MD, Weber TR, Grosfeld J. Preoperative evaluation of pediatric abdominal tumors by computerized tomography.J Pediatr Surg 1984;19:273.
Gattani A, Chesser MR, Cuttner J, Bruckner HW. Serial assays of CA-125, CA-19-9, and CEA in newly treated patients with pancreatic cancer [Abstract].Proc Annu Meet Am Assoc Cancer Res 1992;33:A1305.
Cruickshank DJ, Terry PB, Fullerton WT. CA125-Response assessment in epithelial ovarian cancer.Int J Cancer 1992;51(1):58–61.
Goldie JH, Coldman AJ. A mathematical model for relating the drug sensitivity of tumors to their spontaneous mutation rate.Cancer Treat Rep 1979;63:1727–33.
Goldie JH, Coldman AJ. Quantitative model for multiple levels of drug resistance in clinical tumors.Cancer Treat Rep 1983;67:923–31.
De Vita VT Jr. Principles of chemotherapy. In: De Vita VTJr Hellman SA, Rosenberg SA, eds.Cancer: principles and practice of oncology. Philadelphia: JB Lippincott, 1985:257–83.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Medary, I., Aronson, D., Cheung, NK.V. et al. Kinetics of primary tumor regression with chemotherapy: Implications for the timing of surgery. Annals of Surgical Oncology 3, 521–525 (1996). https://doi.org/10.1007/BF02306083
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02306083