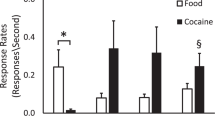
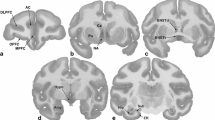
Abstract
The 2-[14C]deoxyglucose method was used to compare the effects of the acute administration of cocaine by two different routes, intravenous and intraperitoneal, on rates of local cerebral glucose utilization in freely moving rats. Doses were initially chosen on the basis of their ability to elicit equivalent increases in locomotor activity during the experimental procedure, and the time of cocaine administration relative to 2-[14C]deoxyglucose infusion was chosen so that the maximal behavioral effect occurred during maximal tracer incorporation. Changes in glucose utilization following the intraperitoneal administration of cocaine (10 mg/kg, 10 min before 2-deoxyglucose infusion) were restricted to the nigrostriatal system and related structures involved in the production of movement. Increased activity was observed in the substantia nigra pars reticulata, globus pallidus, and sensorimotor cortex. In contrast, intravenous cocaine administration (1 mg/kg, 2 min before tracer infusion) produced more widespread changes in rates of glucose utilization including portions of both the mesocorticolimbic and nigrostriatal systems. Areas in which metabolic activity was altered included the caudate-putamen, globus pallidus, substantia nigra pars reticulata, sensorimotor cortex, olfactory tubercle, nucleus accumbens, and medial prefrontal cortex. Both intravenous and intraperitoneal cocaine produced similar increases in locomotor activity. Additional studies indicated that the absence of metabolic activation in the mesocorticolimbic system following acute intraperitoneal cocaine was not the result of the specific dose chosen or the length of time between cocaine administration and radiotracer infusion, as no changes in metabolic activity in mesocorticolimbic structures were evident when these parameters were varied. The cerebral metabolic effects of acute intravenous and intraperitoneal cocaine administration are significantly different. These data indicate that pharmacokinetic variables are important determinants of the functional response to cocaine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Balster RL, Schuster CR (1973) Fixed-interval schedule of cocaine reinforcement: effect of dose and infusion duration. J Exp Anal Behav 20:119–129
Bedford JA, Lovell DK, Turner CE, Elsolly MA, Wilson MC (1980) The anorexic and actometric effects of cocaine and two coca extracts. Pharmacol Biochem Behav 13:403–408
Bradberry CW, Roth RH (1989) Cocaine increases extracellular dopamine in rat nucleus accumbens and ventral tegmental area as shown by in vivo microdialysis. Neurosci Lett 103:97–102
Chow MJ, Ambre JJ, Ruo TI, Atkinson AJ Jr, Bowsher DJ, Fischman MW (1985) Kinetics of cocaine distribution, elimination, and chronotropic effects. Clin Pharmacol Ther 38:318–324
Crane AM, Porrino LJ (1989) Adaptation of the quantitative 2-[14C]deoxyglucose method for use in freely moving rats. Brain Res 499:87–92
Dow-Edwards DL, Fico TA, Osman M, Gamagaris Z, Hutchings DE (1989) Comparison of oral and subcutaneous routes of cocaine administration on behavior, plasma drug concentration and toxicity in female rats. Pharmacol Biochem Behav 33:167–173
Downs DA, Miller LE, Wiley JN, Johnston DE (1980) Oral vs parenteral drug effects of schedule-controlled behavior in rhesus monkeys. Life Sci 26:1163–1168
Fischman MW (1988) Behavioral pharmacology of cocaine. J Clin Psychiatry 49:7–10
Fischman MW, Schuster CR, Rosnekov L, Shick JFE, Krasnegor NA, Fennel W, Freedman DX (1976) Cardiovascular and subjective effects of intravenous cocaine administration in humans. Arch Gen Psychiatry 33:983–990
Foltin RW, Woolverton WL, Schuster CR (1983) Effects of psychomotor stimulants, alone and in pairs, on milk drinking in the rat after intraperitoneal and intragastric administration. J Pharmacol Exp Ther 226:411–418
Heikkila RE, Cabbot FS, Manzino L, Duvoisin RC (1979) Rotational behavior induced by cocaine analogs in rats with unilateral 6-hydroxydopamine lesions of the substantia nigra: dependence upon dopamine uptake inhibition. J Pharmacol Exp Ther 211:189–194
Hertting G, Axelrod J, Whitby LG (1961) Effect of drugs on the uptake and metabolism of3H-norepinephrine. J Pharmacol Exp Ther 134:146–153
Javaid JI, Fischman MW, Schuster CR, Dekirmenjian H, Davis JM (1978) Cocaine plasma concentration: relation to physiological and subjective effects in humans. Science 202:227–228
Johanson C-E, Fischman MW (1989) The pharmacology of cocaine related to its abuse. Pharmacol Rev 41:3–52
Koob GF, Goeders NE (1989) Neuroanatomical substrates of drug self-administration. In: Liebman JM, Cooper SJ (eds) The neuropharmacological basis of reward. Clarendon, Oxford, pp 139–156
Kuczenski R, Segal DS, Aizenstein ML (1991) Amphetamine, cocaine, and fencamfamine: relationship between locomotor and stereotypy response profiles and caudate and accumbens dopamine dynamics. J Neurosci 11:2703–2712
Lau CE, Imam A, Ma F, Falk JL (1991) Acute effects of cocaine on spontaneous and discriminative motor functions: relation to route of administration and pharmacokinetics. J Pharmacol Exp Ther 257:444–456
London ED, Wilkerson G, Goldberg SR, Risner ME (1986) Effects of L-cocaine on local cerebral glucose utilization in the rat. Neurosci Lett 68:73–78
Moore KE, Chiueh CC, Zeldes G (1977) Release of neurotransmitters from the brain in vivo by amphetamine, methylphenidate and cocaine. In: Ellinwood EH, Kilbey MM (eds) Cocaine and other stimulants. Plenum, New York, pp 143–160
Nayak PK, Misra AL, Mulé (1976) Physiological disposition and biotransformation of [3H]cocaine in acutely and chronically treated rats. J Pharmacol Exp Ther 196:556–569
Nomikos GG, Spyraki C (1988) Cocaine-induced place conditioning: importance of route of administration and other procedural variables. Psychopharmacology 94:119–125
Pan H-T, Menacherry S, Justice JB Jr (1991) Differences in the pharmacokinetics of cocaine in naive and cocaine-experienced rats. J Neurochem 56:1299–1306
Paxinos G, Watson C (1982) The rat brain in stereotaxic coordinates. Academic, New York
Pettit HO, Justice HB Jr (1989) Dopamine in the nucleus accumbens during cocaine self-administration as studied by in vivo microdialysis. Pharmacol Biochem Behav 34:899–904
Pettit HO, Pan H-T, Parsons LH, Justice JB Jr (1990) Extracellular concentrations of cocaine and dopamine are enhanced during chronic cocaine administration. J Neurochem 55:798–804
Porrino LJ, Domer FR, Crane AM, Sokoloff L (1988) Selective alterations in cerebral metabolism within the mesocorticolimbic dopaminergic system produced by acute cocaine administration in rats. Neuropsychopharmacology 1:109–118
Resnick RB, Kestenbaum RS, Schwartz LK (1977) Acute systemic effects of cocaine in man: a controller study by intranasal and intravenous routes. Science 195:696–698
Ritchie JM, Greene NM (1980) Local anesthetics. In: Goodman LS, Gilman A (eds) The pharmacological basis of therapeutics. Macmillan, Toronto, pp 300–320
Roberts DCS, Corcoran ME, Fibiger HC (1977) On the role of ascending catecholaminergic systems in intravenous self-administration of cocaine. Pharmacol Biochem Behav 6:615–620
Roberts DCS, Koob GF, Klonoff P, Fibiger HC (1980) Extinction and recovery of cocaine self-administration following 6-hydroxydopamine lesions of the nucleus accumbens. Pharmacol Biochem Behav 12:781–878
Ross SF, Renyi AL (1967) Inhibition of the uptake of tritiated catecholamines by antidepressants and related agents. Eur J Pharmacol 2:181–186
Sharkey J, McBean DE, Kelly PAT (1991) Acute cocaine administration: effects on local cerebral blood flow and metabolic demand in the rat. Brain Res 548:310–314
Shuster L (1992) Pharmacokinetics, metabolism and disposition of cocaine. In: Lakowski JM, Galloway MP, White FJ (eds) Cocaine: pharmacology, physiology, and clinical strategies. CRC Press, Boca Raton, pp 1–14
Sokoloff L, Reivich M, Kennedy C, DesRosiers MH, Patlak CS, Pettigrew KD, Sakurada O, Shinohara M (1977) The [14C]deoxyglucose method for the measurement of local cerebral glucose utilization: theory, procedure and normal values in the conscious and anesthetized albino rat. J Neurochem 28:897–916
Van Dyke C, Barash PG, Jatlow P, Byck R (1976) Cocaine plasma concentrations after intranasal application in man. Science 191:859–861
Van Dyke C, Jatlow PF, Ungerer J, Barash PG, Byck R. (1978) Oral cocaine: plasma concentrations and central effects. Science 200:211–213
Vee GL, Fink GB, Constantine GH Jr (1983) Anorexic activity of cocaine and coca extract in naive and cocaine tolerant rats. Pharmacol Biochem Behav 18:515–517
Wiggins RC, Rolsten C, Ruiz B, Davis CM (1989) Pharmacokinetics of cocaine: basic studies of route, dosage, pregnancy and lactation. Neurotoxicology 10:367–382
Wilkinson P, Van Dyke C, Jatlow P, Barash P, Byck R (1980) Intranasal and oral cocaine kinetics. Clin Pharmacol Ther 27:386–394
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Porrino, L.J. Functional consequences of acute cocaine treatment depend on route of administration. Psychopharmacology 112, 343–351 (1993). https://doi.org/10.1007/BF02244931
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02244931