Abstract
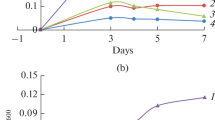
The reaction of guanidine hydrochloride with cyanoacetaldehyde gives high yields (40–85%) of 2,4-diaminopyrimidine under the concentrated conditions of a drying lagoon model of prebiotic synthesis, in contrast to the low yields previously obtained under more dilute conditions. The prebiotic source of cyanoacetaldehyde, cyanoacetylene, is produced from electric discharges under reducing conditions. The effect of pH and concentration of guanidine hydrochloride on the rate of synthesis and yield of diaminopyrimidine were investigated, as well as the hydrolysis of diaminopyrimidine to cytosine, isocytosine, and uracil. Thiourea also reacts with cyanoacetaldehyde to give 2-thiocytosine, but the pyrimidine yields are much lower than with guanidine hydrochloride or urea. Thiocytosine hydrolyzes to thiouracil and cytosine and then to uracil. This synthesis would have been a significant prebiotic source of 2-thiopyrimidines and 5-substituted derivatives of thiouracil, many of which occur in tRNA. The applicability of these results to the drying lagoon model of prebiotic synthesis was tested by dry-down experiments where dilute solutions of cyanoacetaldehyde, guanidine hydrochloride, and 0.5m NaCl were evaporated over varying periods of time. The yields of diaminopyrimidine varied from 1 to 7%. These results show that drying lagoons and beaches may have been major sites of prebiotic syntheses.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- DAP:
-
diaminopyrimidine
- Cy:
-
cyanoacetaldehyde
- C:
-
cytosine
- IC:
-
isocytosine
- U:
-
uracil
- G:
-
guanidine
- GH+ :
-
guanidinium
- G·HCl:
-
guanidinium chloride
- SC:
-
2-thiocytosine
- SU:
-
2-thiouracil
References
Elvers B et al. (eds) Ullmann's encyclopedia of industrial chemistry, vol A12. 5th ed. VHC, New York, p 546
Fabbrizzi L, Micheloni M, Paoletti P, Schwarzenbach G (1977) Protonation process of unusual exothermicity. J Am Chem Soc 99:5574–5576
Ferris JP, Sanchez RA, Orgel LE (1968) Studies in prebiotic synthesis III. Synthesis of pyrimidines from cyanoacetylene and cyanate. J Mol Biol 33:693–704
Ferris JP, Goldstein G, Beaulieu DJ (1970) Chemical evolution IV. An evaluation of cyanovinyl phosphate as a prebiotic phosphorylating agent. J Am Chem Soc 92:6598–6603
Ferris JP, Zamek OS, Altbuch AM, Freiman H (1974) Chemical evolution XVIII. Synthesis of pyrimidines from guanidine and cyanoacetaldehyde. J Mol Evol 3:301–309
Fuller WD, Sanchez RA, Orgel LE (1972) Studies in prebiotic synthesis VII. Solid-state synthesis of purine nucleasides. J Mol Evol 1:249–257
Fox SW, Dose K (1972) Molecular evolution and the origin of life. WH Freeman, San Francisco
Frost AA, Pearson RG (1961) Kinetics and mechanism. John Wiley, New York, pp 166–169
Garrett ER, Tsau J (1972) Solvolyses of cytosine and cytidine. J Pharm Sci 61:1052–1061
Keefe AD, Newton GL, Miller SL (1995) A possible prebiotic synthesis of pantetheine, a precursor to coenzyme A. Nature 373:683–685
Kirk RE, Othmer DF (1951) Encyclopedia of chemical technology, vol. 7. Interscience Encyclopedia, New York, p 326
Limbach PA, Crain PF, McCloskey JA (1994) Summary: the modified nucleosides of RNA. Nucleic Acids Res 22:2183–2196
Lohrmann R (1972) Formation of urea and guanidine by irradiation of ammonium cyanide. J Mol Evol 1:263–269
Lowe CU, Rees MW, Markham R (1963) Synthesis of complex organic compounds from simple precursors: formation of amino-acids, amino-acid polymers, fatty acids and purines from ammonium cyanide. Nature 199:219–222
Miller SL (1957) The mechanism of synthesis of amino acids by electric discharges. Biochim Biophys Acta 23:480–489
Miller SL, Orgel LE (1974) The origins of life on the earth. Prentice-Hall, Englewood Cliffs, NJ, pp 129–134
Piccirilli JA, Krauch T, Moroney SE, Benner SA (1990) Enzymatic incorporation of a new base pair into DNA and RNA extends the genetic alphabet. Nature 343:33–37
Reid EE (1963) Organic chemistry of bivalent sulfur. vol. 5. Chemical Publishing Co. Inc., New York, pp 11–17
Robertson MP, Miller SL (1995a) An efficient prebiotic synthesis of cytosine and uracil. Nature 375:772–774
Robertson MP, Miller SL (1995b) Prebiotic synthesis of 5-substituted uracils: a bridge between the RNA world and the DNA-protein world. Science 268:702–705
Sanchez RA, Ferris JP, Orgel LE (1966) Cyanoacetylene in prebiotic synthesis. Science 154:784–785
Shapiro R (1995) The prebiotic role of adenine: a critical analysis. Orig Life Evol Biosph 25:83–98
Shapiro R, Klein RS (1966) The deamination of cytidine and cytosine by acidic buffer solutions. Mutagenic implications. Biochemistry 5:2358–2362
Shaw WHR, Walker DG (1958) Kinetic studies of thiourea derivatives. IV. The methylated thioureas. Conclusions. J Am Chem Soc 80:5337–5342
Shnidman L (1933) The solubility of thiourea in water, methanol, and ethanol. J Phys Chem 37:693–700
Switzer C, Moroney SE, Benner SA (1989) Enzymatic incorporation of a new base pair into DNA and RNA. J Am Chem Soc 111:8322–8323
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Robertson, M.P., Levy, M. & Miller, S.L. Prebiotic synthesis of diaminopyrimidine and thiocytosine. J Mol Evol 43, 543–550 (1996). https://doi.org/10.1007/BF02202102
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02202102