Abstract
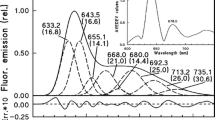
77 K fluorescence spectra of bean (Phaseolus vulgaris cv Commodore) leaves and cotyledons show the presence of active and inactive protochlorophyllides. The first detected product of the protochlorophyllide photoreduction is the chlorophyllide emitting fluorescence at 690 nm (C690) which is observed one day (leaves) and three days (cotyledons) after sowing. In cotyledons, C690 undergoes the ‘rapid’ and ‘Shibata’ shifts; in leaves, these spectral changes are age-dependent. In order to characterize the formation of C690, we have recorded 298 K fluorescence kinetics at 690 nm and the corresponding absorbance kinetics at 440 nm. The amplitude of the variations of both kinetics increases with the sample age. The absorbance and fluorescence kinetics can be modelized as a monoexponential law. The rate constant of the absorbance and fluorescence kinetics does not significantly change during the studied period (except for old cotyledons). The results presented in this paper give evidences for a low energy transfer between pigments during the photoreduction of protochlorophyllide at room temperature.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Ax:
-
absorbance intensity measured at x nm
- Chlide:
-
chlorophyll(ide)
- A.U.:
-
arbitrary unit
- Cx:
-
chlorophyll(ide) fluorescing at x nm at 77 K
- Fx:
-
fluorescence intensity measured at x nm
- PCR:
-
NADPH:protochlorophyllide oxidoreductase (EC 1.6.99.1)
- Pchlide:
-
protochlorophyllide
- Px:
-
protochlorophyllide fluorescing at x nm at 77K
References
Adamson H, Leenon M, Ou KL, Packer N and Walmsley J (1990) Evidence for a light-independent chlorophyll biosynthetic pathway in angiosperm seeds germinated in darkness. In: Baltscheffsky M (ed) Current Research in Photosynthesis, Vol III, pp 607–609. Kluwer Academic Publishers, Dordrecht, Boston, London
Bereza B and Dujardin E (1981) Difference absorption changes in the visible region during and after photoreduction of protochlorophyllide. In: Akoyunoglou G (ed) Photosynthesis. Chloroplast Development, Vol V, PP 15–20. Balaban International Science Services, Philadelphia, PA
Bertrand M, Bereza B and Dujardin E (1988) Evidence for photoreduction of NADP+ in a suspension of lysed plastids from etiolated leaves. Z Naturforsch 43c: 443–448
Boardman NK (1962) Studies on a protochlorophyll-protein complex. II. The photoconversion of protochlorophylla in isolated complex. Biochim Biophys Acta 64: 279–293
Boardman NK (1966) Protochlorophyll. In: Vernon LP and Seely GR (eds) The Chlorophyll, pp 437–479. Academic Press, New York
Böddi B (1990) Spectral properties and molecular structure of protochlorophyll, protochlorophyllide and chlorophylla forms in model and in isolated etioplasts fragments. In: Baltscheffsky M (ed) Current Research in Photosynthesis, Vol III, pp 835–842. Kluwer Academic Publishers, Dordrecht, Boston, London
Böddi B, Ryberg M and Sundqvist C (1992) Identification of four universal protochlorophyllide forms in dark-grown leaves by analyses of the 77 K fluorescence emission spectra. J Photochem Photobiol, B Biol 12: 389–401
Brouers M, Kuyper K and Sironval C (1972) The reduction of protochlorophyllide into chlorophyllide. V. Demonstration of energy transfer inside the P688–676 units. Photosynthetica 6: 169–176
Brouers M and Sironval C (1974) Evidence for energy transfer from protochlorophyllide to chlorophyllide in leaves treated with δ-aminolevulinic acid. Plant Sci Lett 2: 67–72
Brouers M and Sironval C (1978) The reduction of protochlorophyllide into chlorophyllide. VII. Relations between energy transfer, 690 nm fluorescence emission, and reduction; a theory. Photosynthetica 12: 399–405
Cachon D and Geneves L (1985) Ultrastructure des cellules cotylédonnaires, des grains protéiques et des cristaux dans les graines sèches dePhaseolus vulgaris L. Annales des Sciences Naturelles Botaniques, Paris, Sér 13, 7: 131–148
Dobek A, Dujardin E, Franck F, Sironval C, Breton J and Roux E (1981) The first events of protochlorophyll(ide) photoreduction investigated in etiolated leaves by means of the fluorescence excited by short, 610 nm laser flashes at room temperature. Photobiochem Photobiophys 2: 35–44
Dujardin E (1975) Energy transfers between the protochloropyll(ide) forms in lyophilized etiolated bean leaves. Photosynthetica 9: 283–287
Dujardin E and Correia M (1979) Long-wavelength absorbing pigment protein complexes as fluorescence quenchers in etiolated leaves illuminated in liquid nitrogen. Photobiochem Photobiophys 1: 25–32
Dujardin E, Mathis P and Kahn A (1990) Soret absorption of the intermediate(s) in protochlorophyllide to chlorophyllide photoreduction trapped at low temperature. Physiol Plant 78: 123–127
Durchan M, Pakshina EV and Lebedev NN (1993) Traces of chlorophylla and the spectral forms of protochlorophyll(ide)a in etiolated cucumber cotyledons. Photosynthetica 28: 567–572
ElHamouri B and Sironval C (1979) A new non-photoreducible protochlorophyll(ide)-protein:P649–642 from cucumber cotyledon; NADPH mediation of its transformation to photoreducible P657–650. FEBS Lett 103: 345–347
ElHamouri B and Sironval C (1980) NADP+/NADPH control of the protochlorophyllide-, chlorophyllide-proteins in cucumber etioplasts. Photobiochem Photobiophys 1: 219–223
Franck F and Strzalka K (1992) detection of the photoactive protochlorophyllide-protein complex in the light during the greening barley. FEBS Lett 309: 73–77
French CS (1960) The chlorophylls in vivo and in vitro. In: Ruhland W (ed) Encyclopediae of Plant Physiology, Vol 1, pp 252–297. Springer Verlag, Berlin
Garnir HP and Monjoie F (1981) Fit of the function 416–1 to data strongly embedded in noise. Nucl Instrum and Methods 190: 333–336
Gassman M and Bogorad L (1967) Studied on the regeneration of protochlorophyllide after a brief illumination of etiolated bean leaves. Plant Physiol 42: 781–784
Goedheer JC and Verhülsdonk CAH (1970) Fluorescence and phototransformation of protochlorophyll with etiolated bean leaves from −196 to + 20 °C. Biochem Biophys Res Commun 39: 260–266
Granick S and Gassman M (1970) Rapid regeneration of protochlorophyllide 650. Plant Physiol 45: 201–205
Harnitschfeger G (1977) The use of fluorescence emission at 77°K in the analysis of the photosynthetic apparatus in higher plants and algae. Adv Bot Res 5: 2–52
Huber DJ and Newman DW (1976) Relationships between lipid changes and plastid ultrastructural changes in senescing and regreening soybean cotyledons. J Exp Bot 27: 490–511
Huner NPA, Öquist G and Sundblad LG (1992) Low temperature measuring artifactual increase in chlorophyll a fluorescence. Plant Physiol 98: 742–752
Inoue Y, Kobayashi T, Ogawa T and Shibata K (1981) A short lived intermediate in the photoconversion of protochlorophyllide to chrophyllidea. Plant Cell Physiol 22: 197–204
Jouy M (1982) Effect of age of etiolated leaves ofPhaseolus vulgaris on the 695 nm fluorescence kinetics during the first irradiation. Photosynthetica 16: 234–238
Jouy M and Sironval C (1979) Quenching of the fluorescence emitted by P695–682 at room temperature in etiolated illuminated leaves. Planta 147: 127–133
Kahn A, Boardman NK and Thorne SW (1970) Energy transfer between protochlorophyllide molecules: Evidence for multiple chromophores in the photoactive protochlorophyllide-protein complex in vivo and in vitro. J Mol Biol 48: 85–101
Kahn A and Nielsen O (1974) Photoconvertible protochlorophyll(ide) 635/650 in vivo: A single species or two species in dynamic equilibrium. Biochim Biophys Acta 333: 409–414
Khandakar K and Bradbeer JW (1988) Primary leaf growth in bean (Phaseolus vulgaris L.). II. Cell and plastid development during growth in darkness and after transfer to illumination at various stages of dark growth. Bangladesh J Bot 17: 173–188
Klein S and Schiff JA (1972) Correlated appearance of prolamellar bodies, protochlorophyll(ide) species, and the Shibata shift during development of bean etioplasts in the dark. Plant Physiol 49: 612–626
Litvin FF and Stadnitshuk IN (1980) Long wavelength forms of chlorophyll precursors in etiolated leaves and in systems of native forms of protochlorophyllide. Fiz Rast 27: 1024–1030 (in Russian)
Madsen A (1963) On the formation of chlorophyll and initiation of photosynthesis in etiolated plants. Photochem Photobiol 2: 93–100
Monjoie FS and Garnir HP (1993) Fit of a sum of exponential function to experimental data points. Computer Physics Communications 74: 1–8
Nielsen OF and Kahn A (1973) Kinetics and quantum yield of photoconversion of protochlorophyll(ide) to chlorophyll(ide)a. Biochim Biophys Acta 229: 117–129
Ogawa M and Konishi M (1979) Kinetics of photoconversion of protochlorophyllide 649 to chlorophyllide 676 at low temperature in etiolated cotyledons ofPharbitis nil. Biochim Biophys Acta 548: 119–127
Oliver RP and Griffiths WT (1982) Pigment-protein complexes of illuminated etiolated leaves. Plant Physiol, 70: 1019–1025
Rebeiz CA, Yaghi M. and Abou-Haidar H (1970) Protochlorophyll biosynthesis in cucumber (Cucumis sativus L.) cotyledons. Plant Physiol 46: 57–63
Rorabacher DB (1991) Statistical treatement for rejection of deviant values. Critical values of Dixon's ‘Q’ parameters and related subrange ratios at 95% confidence level. Anal Chem 63: 139–146
Savchenko GE, Abramchik LM, Klyuchareva EA and Chaika MT (1990) NDPH:protochlorophyllide oxidoreductase in barley (Hordeum vulgare) seedlings. In. Baltscheffsky M (ed) Current Research in Photosynthesis, Vol III, pp 819–822. Kluwer Academic Publishers, Dordrecht, Boston, London
Schoefs B and Franck F (1990) Photoreduction of the protochlorophyllide into chlorophyllide in etiolated leaves and cotyledons fromPhaseolus vulgaris cv Commodore. In: Baltscheffsky M (ed) Current Research in Photosynthesis, Vol III, pp 755–758. Kluwer Academic Publishers, Dordrecht, Boston, London
Schoefs B and Franck F (1993) Photoreduction of protochlorophyllide to chlorophyllide in 2-day-old dark-grown bean (Phaseolus vulgaris cv Commodore) leaves. A comparison with 10-day-old (etiolated) leaves. J Exp Bot 44: 1053–1057
Schoefs B, Bertrand M and Franck F (1992a) On the formation of chlorophyllide after phototransformation of protochlorophyllide in 2-day old bean leaves. Comparison with etiolated leaves. In: Argyroudi-Akoyunoglou JH (ed) Regulation of Chloroplast Biogenesis, pp 233–237. Plenum Publishing Company, New York
Schoefs B, Bertrand M and Franck F (1992b) Plant greening: The case of bean leaves illuminated shortly after the germination. Photosynthetica 24: 497–504
Schoefs B, Bertrand M and Franck F (1993) Kinetics of the photoreduction of protochlorophyllide to chlorophyllide in leaves ofPhaseolus vulgaris cv Commodore. In: Theophanides T, Anastassopoulou J and Fotopoulos N (eds) 5th International Conference on the Spectroscopy of the Biological Molecules, pp 303–304. Kluwer Academic Publishers, Dordrecht, Boston, London
Shibata K (1957) Spectroscopic studies on chlorophyll formation in intact leaves. J Biochem 44: 147–173.
Shlyk AA, Savchenko GY and Averina VG (1969) Investigation of the kinetics of photoreduction of protochlorophyllide in green leaves by the spectrofluorographic method. Biofizika 14: 119–129
Sironval C and Brouers M (1970) The reduction of protochlorophyllide into chlorophyllide. II. The temperature dependence of the P657–647 → P688–676 phototransformation. Photosynthetica 4: 38–47
Sironval C and Brouers M (1980) The reduction of protochlorophyllide into chlorophyllide. VIII. The theory of the transfer units. Photosynthetica 14: 213–221
Sironval C and Kuyper Y (1972) The reduction of protochlorophyllide into chlorophyllide. IV. The nature of the intermediate P688–676 species. Photosynthetica 6: 254–275
Sironval C, Bronchart R, Michel JM, Brouers M and Kuyper Y (1968a) Structure macromoléculaire et activités photochimiques des lamelles plastidiales (essais). Bull Soc Physiol Végét 14: 195–225
Sironval C, Brouers M, Michel JM and Kuiper Y (1968b) The reduction of the protochlorophyllide into chlorophyllide. I. Kinetics of the P657–647 phototransformation. Photosynthetica 2: 268–287
Sironval C, Franck F, Gysemberg R, Bereza B and Dujardin E (1984) The Franck-Inoue chlorophyllide microcycle II in vivo and in vitro. In: Sironval C and Brouers M (eds) Protochlorophyllide Photoreduction and Greening, pp 197–222. Martinus Nijhoff/Dr. W. Junk Publishers, The Hague
Smith JHC and Benitez A (1954) The effect of temperature on the conversion of protochlorophyll to chlorophylla in etiolated barley leaves. Plant Physiol 29: 135–143
Thorne SW (1971) The greening of etiolated bean leaves. I. The initial photoconversion process. Biochim Biophys Acta 226: 113–127
Thorne SW and Boardman NK (1972) The kinetics of the photoconversion of protochlorophyllide in etiolated bean leaves. Biochim Biophys Acta 267: 104–110
VanBochove AC, Griffiths WT and vanGrondele R (1984) The primary reaction in the photoreduction of protochlorophyllide. A nanosecond fluorescence study. In: Sironval C and Brouers M (eds) Protochlorophyllide Photoreduction and Greening, pp 113–125. Martinus Nijhoff/Dr. W. Junk Publishers, The Hague
Vaughan GD and Sauer K (1974) Energy transfer from protochlorophyllide to phlorophyllide during the photoconversion of etiolated bean holochrome. Biochim Biophys Acta 347: 383–394
Virgin HI (1955) The conversion of protochlorophyll to chlorophylla in continuous and intermittent light. Physiol Plant 8: 389–403
Virgin HI (1981) The physical state of protochlorophyll(ide) in plants. Annu Rev Plant Physiol 32: 451–463
Webster BD and Leopold AC (1977) The ultrastructure of dry and imbibed cotyledons of soybean. Amer J Bot 64: 1286–1293
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schoefs, B., Garnir, HP. & Bertrand, M. Comparison of the photoreduction of protochlorophyllide to chlorophyllide in leaves and cotyledons from dark-grown bean as a function of age. Photosynth Res 41, 405–417 (1994). https://doi.org/10.1007/BF02183043
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02183043